Selective estrogen receptor modulators (SERMs), also known as estrogen receptor agonists/antagonists (ERAAs), are a class of drugs that act on estrogen receptors (ERs). Compared to pure ER agonists–antagonists, SERMs are more tissue-specific, allowing them to selectively inhibit or stimulate estrogen-like action in various tissues.

Estrogen receptors (ERs) are proteins found in cells that function as receptors for the hormone estrogen (17β-estradiol). There are two main classes of ERs. The first includes the intracellular estrogen receptors, namely ERα and ERβ, which belong to the nuclear receptor family. The second class consists of membrane estrogen receptors (mERs), such as GPER (GPR30), ER-X, and Gq-mER, which are primarily G protein-coupled receptors. This article focuses on the nuclear estrogen receptors.

Histone acetyltransferase p300 also known as p300 HAT or E1A-associated protein p300 also known as EP300 or p300 is an enzyme that, in humans, is encoded by the EP300 gene. It functions as histone acetyltransferase that regulates transcription of genes via chromatin remodeling by allowing histone proteins to wrap DNA less tightly. This enzyme plays an essential role in regulating cell growth and division, prompting cells to mature and assume specialized functions (differentiate), and preventing the growth of cancerous tumors. The p300 protein appears to be critical for normal development before and after birth.

Proline-, glutamic acid- and leucine-rich protein 1 (PELP1) also known as modulator of non-genomic activity of estrogen receptor (MNAR) and transcription factor HMX3 is a protein that in humans is encoded by the PELP1 gene. is a transcriptional corepressor for nuclear receptors such as glucocorticoid receptors and a coactivator for estrogen receptors.
In molecular biology and genetics, transcription coregulators are proteins that interact with transcription factors to either activate or repress the transcription of specific genes. Transcription coregulators that activate gene transcription are referred to as coactivators while those that repress are known as corepressors. The mechanism of action of transcription coregulators is to modify chromatin structure and thereby make the associated DNA more or less accessible to transcription. In humans several dozen to several hundred coregulators are known, depending on the level of confidence with which the characterisation of a protein as a coregulator can be made. One class of transcription coregulators modifies chromatin structure through covalent modification of histones. A second ATP dependent class modifies the conformation of chromatin.

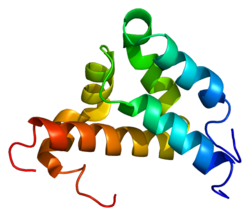
Estrogen receptor alpha (ERα), also known as NR3A1, is one of two main types of estrogen receptor, a nuclear receptor that is activated by the sex hormone estrogen. In humans, ERα is encoded by the gene ESR1.
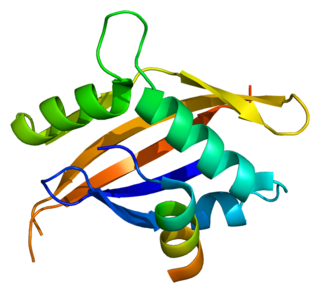
The nuclear receptor coactivator 1 (NCOA1), also called steroid receptor coactivator-1 (SRC-1), is a transcriptional coregulatory protein that contains several nuclear receptor–interacting domains and possesses intrinsic histone acetyltransferase activity. It is encoded by the gene NCOA1.
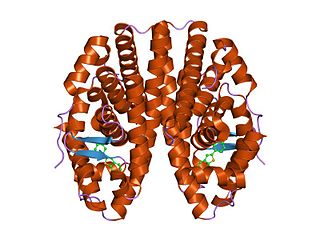
The nuclear receptor coactivator 2 also known as NCoA-2 is a protein that in humans is encoded by the NCOA2 gene. NCoA-2 is also frequently called glucocorticoid receptor-interacting protein 1 (GRIP1), steroid receptor coactivator-2 (SRC-2), or transcriptional mediators/intermediary factor 2 (TIF2).

Bert W. O'Malley is an endocrinologist from the United States. He was born in 1936 in the Garfield section of Pittsburgh, Pennsylvania. He received his early education at Catholic primary schools and Central Catholic High School, before pursuing higher education at the University of Pittsburgh, where he completed both his undergraduate and medical studies, graduating first in his class. It was here that he met Sally, who would become his wife and lifelong partner. The couple went on to have four children.

Retinoid X receptor alpha (RXR-alpha), also known as NR2B1 is a nuclear receptor that in humans is encoded by the RXRA gene.

AKT2, also known as RAC-beta serine/threonine-protein kinase, is an enzyme that in humans is encoded by the AKT2 gene. It influences metabolite storage as part of the insulin signal transduction pathway.

Signal transducer and activator of transcription 5A is a protein that in humans is encoded by the STAT5A gene. STAT5A orthologs have been identified in several placentals for which complete genome data are available.
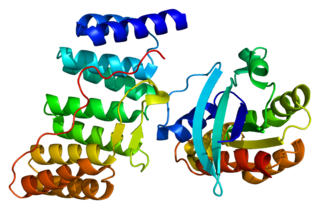
Ras-related C3 botulinum toxin substrate 3 (Rac3) is a G protein that in humans is encoded by the RAC3 gene. It is an important component of intracellular signalling pathways. Rac3 is a member of the Rac subfamily of the Rho family of small G proteins. Members of this superfamily appear to regulate a diverse array of cellular events, including the control of cell growth, cytoskeletal reorganization, and the activation of protein kinases.

Mediator of RNA polymerase II transcription subunit 1 also known as DRIP205 or Trap220 is a subunit of the Mediator complex and is a protein that in humans is encoded by the MED1 gene. MED1 functions as a nuclear receptor coactivator.
Probable ATP-dependent RNA helicase DDX5 also known as DEAD box protein 5 or RNA helicase p68 is an enzyme that in humans is encoded by the DDX5 gene.

Scaffold attachment factor B, also known as SAFB, is a gene with homologs that have been studied in humans and mice.

Nuclear receptor coactivator 6 is a protein that in humans is encoded by the NCOA6 gene.

Steroid receptor RNA activator 1 also known as steroid receptor RNA activator protein (SRAP) is a protein that in humans is encoded by the SRA1 gene. The mRNA transcribed from the SRA1 gene is a component of the ribonucleoprotein complex containing NCOA1. This functional RNA also encodes a protein.

Tripartite motif-containing 24 (TRIM24) also known as transcriptional intermediary factor 1α (TIF1α) is a protein that, in humans, is encoded by the TRIM24 gene.
Nuclear receptor coregulators are a class of transcription coregulators that have been shown to be involved in any aspect of signaling by any member of the nuclear receptor superfamily. A comprehensive database of coregulators for nuclear receptors and other transcription factors was previously maintained at the Nuclear Receptor Signaling Atlas website which has since been replaced by the Signaling Pathways Project website.