 | |
 | |
| Names | |
|---|---|
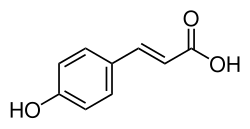
| Preferred IUPAC name (2E)-3-(4-Hydroxyphenyl)prop-2-enoic acid | |
| Other names (E)-3-(4-Hydroxyphenyl)-2-propenoic acid (E)-3-(4-Hydroxyphenyl)acrylic acid para-Coumaric acid 4-Hydroxycinnamic acid trans-4-Hydroxycinnamic acid β-(4-Hydroxyphenyl)acrylic acid | |
| Identifiers | |
3D model (JSmol) | |
| 2207383 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.116.210 |
| EC Number |
|
| 2245630 | |
| KEGG | |
PubChem CID | |
| UNII | |
CompTox Dashboard (EPA) | |
| |
| |
| Properties | |
| C9H8O3 | |
| Molar mass | 164.160 g·mol−1 |
| Melting point | 210 to 213 °C (410 to 415 °F; 483 to 486 K) |
| Hazards | |
| GHS labelling: | |
 | |
| Danger | |
| H315, H319, H335 | |
| P261, P264, P271, P280, P302+P352, P305+P351+P338 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
p-Coumaric acid is an organic compound with the formula HOC6H4CH=CHCO2H. It is one of the three isomers of coumaric acid. It is a white solid that is only slightly soluble in water but very soluble in ethanol and diethyl ether.