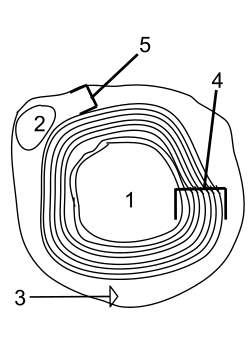
An axon, or nerve fiber, is a long, slender projection of a nerve cell, or neuron, in vertebrates, that typically conducts electrical impulses known as action potentials away from the nerve cell body. The function of the axon is to transmit information to different neurons, muscles, and glands. In certain sensory neurons, such as those for touch and warmth, the axons are called afferent nerve fibers and the electrical impulse travels along these from the periphery to the cell body and from the cell body to the spinal cord along another branch of the same axon. Axon dysfunction can cause many inherited and acquired neurological disorders which can affect both the peripheral and central neurons. Nerve fibers are classed into three types – group A nerve fibers, group B nerve fibers, and group C nerve fibers. Groups A and B are myelinated, and group C are unmyelinated. These groups include both sensory fibers and motor fibers. Another classification groups only the sensory fibers as Type I, Type II, Type III, and Type IV.

Myelin is a lipid-rich material that surrounds nerve cell axons to insulate them and increase the rate at which electrical impulses are passed along the axon. The myelinated axon can be likened to an electrical wire with insulating material (myelin) around it. However, unlike the plastic covering on an electrical wire, myelin does not form a single long sheath over the entire length of the axon. Rather, myelin sheaths the nerve in segments: in general, each axon is encased with multiple long myelinated sections with short gaps in between called nodes of Ranvier.

A nerve is an enclosed, cable-like bundle of nerve fibers in the peripheral nervous system.
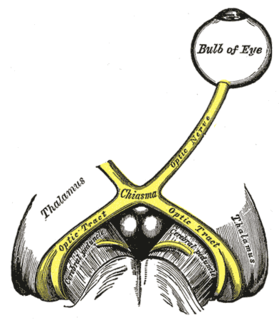
In neuroanatomy, the optic nerve, also known as the second cranial nerve, cranial nerve II, or simply CN II, is a paired cranial nerve that transmits visual information from the retina to the brain. In humans, the optic nerve is derived from optic stalks during the seventh week of development and is composed of retinal ganglion cell axons and glial cells; it extends from the optic disc to the optic chiasma and continues as the optic tract to the lateral geniculate nucleus, pretectal nuclei, and superior colliculus.
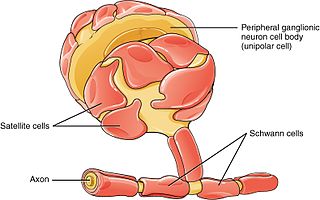
Schwann cells or neurolemmocytes are the principal glia of the peripheral nervous system (PNS). Glial cells function to support neurons and in the PNS, also include satellite cells, olfactory ensheathing cells, enteric glia and glia that reside at sensory nerve endings, such as the Pacinian corpuscle. The two types of Schwann cells are myelinating and nonmyelinating. Myelinating Schwann cells wrap around axons of motor and sensory neurons to form the myelin sheath. The Schwann cell promoter is present in the downstream region of the human dystrophin gene that gives shortened transcript that are again synthesized in a tissue-specific manner.
Nervous tissue, also called neural tissue, is the main tissue component of the nervous system. The nervous system regulates and controls body functions and activity. It consists of two parts: the central nervous system (CNS) comprising the brain and spinal cord, and the peripheral nervous system (PNS) comprising the branching peripheral nerves. It is composed of neurons, also known as nerve cells, which receive and transmit impulses, and neuroglia, also known as glial cells or glia, which assist the propagation of the nerve impulse as well as provide nutrients to the neurons.

A motor nerve is a nerve located in the central nervous system (CNS), usually the spinal cord, that sends motor signals from the CNS to the muscles of the body. This is different from the motor neuron, which includes a cell body and branching of dendrites, while the nerve is made up of a bundle of axons. Motor nerves act as efferent nerves which carry information out from the CNS to muscles, as opposed to afferent nerves, which send signals from sensory receptors in the periphery to the CNS. Efferent nerves can also connect to glands or other organs/issues instead of muscles. In addition, there are nerves that serve as both sensory and motor nerves called mixed nerves.

Oligodendrocytes, or oligodendroglia, are a type of neuroglia whose main functions are to provide support and insulation to axons in the central nervous system of jawed vertebrates, equivalent to the function performed by Schwann cells in the peripheral nervous system. Oligodendrocytes do this by creating the myelin sheath. A single oligodendrocyte can extend its processes to 50 axons, wrapping approximately 1 μm of myelin sheath around each axon; Schwann cells, on the other hand, can wrap around only one axon. Each oligodendrocyte forms one segment of myelin for several adjacent axons.

Glia, also called glial cells(gliocytes) or neuroglia, are non-neuronal cells in the central nervous system and the peripheral nervous system that do not produce electrical impulses. They maintain homeostasis, form myelin in the peripheral nervous system, and provide support and protection for neurons. In the central nervous system, glial cells include oligodendrocytes, astrocytes, ependymal cells, and microglia, and in the peripheral nervous system they include Schwann cells and satellite cells.
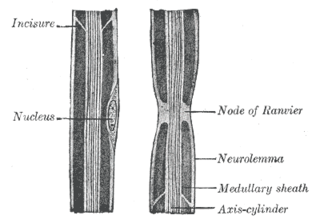
In neuroscience and anatomy, Nodes of Ranvier, also known as myelin-sheath gaps, occur along a myelinated axon where the axolemma is exposed to the extracellular space. Nodes of Ranvier are uninsulated and highly enriched in ion channels, allowing them to participate in the exchange of ions required to regenerate the action potential. Nerve conduction in myelinated axons is referred to as saltatory conduction due to the manner in which the action potential seems to "jump" from one node to the next along the axon. This results in faster conduction of the action potential.

Wallerian degeneration is an active process of degeneration that results when a nerve fiber is cut or crushed and the part of the axon distal to the injury degenerates. A related process of dying back or retrograde degeneration known as 'Wallerian-like degeneration' occurs in many neurodegenerative diseases, especially those where axonal transport is impaired such as ALS and Alzheimer's disease. Primary culture studies suggest that a failure to deliver sufficient quantities of the essential axonal protein NMNAT2 is a key initiating event.
Axonotmesis is an injury to the peripheral nerve of one of the extremities of the body. The axons and their myelin sheath are damaged in this kind of injury, but the endoneurium, perineurium and epineurium remain intact. Motor and sensory functions distal to the point of injury are completely lost over time leading to Wallerian degeneration due to ischemia, or loss of blood supply. Axonotmesis is usually the result of a more severe crush or contusion than neurapraxia.

Myelin incisures, are small pockets of cytoplasm left behind during the Schwann cell myelination process.
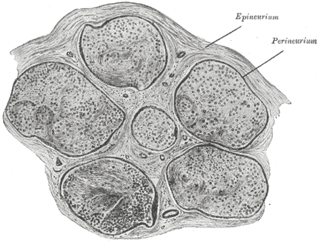
The perineurium is a protective sheath that surrounds a nerve fascicle. This bundles together axons targeting the same anatomical location. The perineurium is composed from fibroblasts.

The endoneurium is a layer of delicate connective tissue around the myelin sheath of each myelinated nerve fiber in the peripheral nervous system. Its component cells are called endoneurial cells. The endoneuria with their enclosed nerve fibers are bundled into groups called nerve fascicles, each fascicle within its own protective sheath called a perineurium. In sufficiently large nerves multiple fascicles, each with its blood supply and fatty tissue, may be bundled within yet another sheath, the epineurium.

Nerve injury is an injury to nervous tissue. There is no single classification system that can describe all the many variations of nerve injuries. In 1941, Seddon introduced a classification of nerve injuries based on three main types of nerve fiber injury and whether there is continuity of the nerve. Usually, however, peripheral nerve injuries are classified in five stages, based on the extent of damage to both the nerve and the surrounding connective tissue, since supporting glial cells may be involved.
Neuroregeneration refers to the regrowth or repair of nervous tissues, cells or cell products. Such mechanisms may include generation of new neurons, glia, axons, myelin, or synapses. Neuroregeneration differs between the peripheral nervous system (PNS) and the central nervous system (CNS) by the functional mechanisms involved, especially in the extent and speed of repair. When an axon is damaged, the distal segment undergoes Wallerian degeneration, losing its myelin sheath. The proximal segment can either die by apoptosis or undergo the chromatolytic reaction, which is an attempt at repair. In the CNS, synaptic stripping occurs as glial foot processes invade the dead synapse.
Myelinogenesis is the formation and development of myelin sheaths in the nervous system, typically initiated in late prenatal neurodevelopment and continuing throughout postnatal development. Myelinogenesis continues throughout the lifespan to support learning and memory via neural circuit plasticity as well as remyelination following injury. Successful myelination of axons increases action potential speed by enabling saltatory conduction, which is essential for timely signal conduction between spatially separate brain regions, as well as provides metabolic support to neurons.
In neurobiology, a mesaxon is a pair of parallel plasma membranes of a Schwann cell. It marks the point of edge-to-edge contact by the Schwann cell encircling the axon. A single Schwann cell of the peripheral nervous system will wrap around and support only one individual axon, while the oligodendrocytes found in the central nervous system can wrap around and support 5-8 axons. Thin unmyelinated axons are often bundled, with several unmyelinated axons to a single mesaxon.
Anti-MAG peripheral neuropathy is a specific type of peripheral neuropathy in which the person's own immune system attacks cells that are specific in maintaining a healthy nervous system. As these cells are destroyed by antibodies, the nerve cells in the surrounding region begin to lose function and create many problems in both sensory and motor function. Specifically, antibodies against myelin-associated glycoprotein (MAG) damage Schwann cells. While the disorder occurs in only 10% of those afflicted with peripheral neuropathy, people afflicted have symptoms such as muscle weakness, sensory problems, and other motor deficits usually starting in the form of a tremor of the hands or trouble walking. There are, however, multiple treatments that range from simple exercises in order to build strength to targeted drug treatments that have been shown to improve function in people with this type of peripheral neuropathy.