
Sulfuric acid or sulphuric acid, known in antiquity as oil of vitriol, is a mineral acid composed of the elements sulfur, oxygen, and hydrogen, with the molecular formula H2SO4. It is a colorless, odorless, and viscous liquid that is miscible with water.
Sulfur oxide refers to many types of sulfur and oxygen containing compounds such as SO, SO2, SO3, S7O2, S6O2, S2O2, etc.
Iron sulfide or Iron sulphide can refer to range of chemical compounds composed of iron and sulfur.
Dibromoethane can refer to either of two isomeric organobromides with the molecular formula C2H4Br2:
Sulfur nitride may refer to a number of sulfur nitrogen compounds:

Ditellurium bromide is the inorganic compound with the formula Te2Br. It is one of the few stable lower bromides of tellurium. Unlike sulfur and selenium, tellurium forms families of polymeric subhalides where the halide/chalcogen ratio is less than 2.
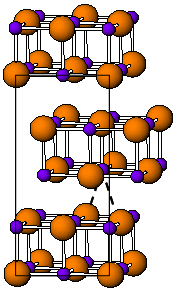
Indium(I) bromide is a chemical compound of indium and bromine. It is a red crystalline compound that is isostructural with β-TlI and has a distorted rock salt structure. Indium(I) bromide is generally made from the elements, heating indium metal with InBr3. It has been used in the sulfur lamp. In organic chemistry, it has been found to promote the coupling of α, α-dichloroketones to 1-aryl-butane-1,4-diones. Oxidative addition reactions with for example alkyl halides to give alkyl indium halides and with NiBr complexes to give Ni-In bonds are known. It is unstable in water decomposing into indium metal and indium tribromide. When indium dibromide is dissolved in water, InBr is produced as a, presumably, insoluble red precipitate, that then rapidly decomposes.
The molecular formula C2H4Br2 (molar mass: 187.86 g/mol, exact mass: 185.8680 u) may refer to:
Tellurium iodide is an inorganic compound with the formula TeI. Two forms are known. Their structures differ from the other monohalides of tellurium. There are three subiodides of tellurium, α-TeI, β-TeI, and Te2I, and one tellurium tetraiodide.
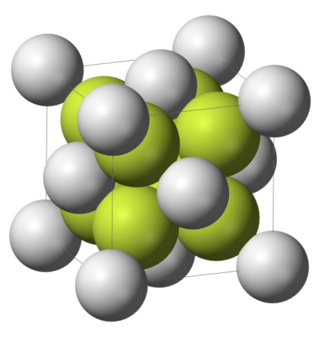
Difluorides are chemical compounds with two fluorine atoms per molecule.

Disulfur dibromide is the inorganic compound with the formula S2Br2. It is a yellow-brown liquid that fumes in air. It is prepared by direct combination of the elements and purified by vacuum distillation. The compound has no particular application, unlike the related sulfur compound disulfur dichloride.

Sulfur dibromide is the chemical compound with the formula SBr2. It is a toxic gas.
Neodymium bromide may refer to:

Xylylene dibromide is an organic compound with the formula C6H4(CH2Br)2. It is an off-white solid that, like other benzyl halides, a strong lachrymator. It is a useful reagent owing to the convenient reactivity of the two C-Br bonds. Two other isomers are known, para- and meta-xylylene dibromide.
Lithium battery may refer to:

Disulfur diiodide is an unstable inorganic chemical compound with the chemical formula S2I2. Its empirical formula is SI. It is a red-brown solid that decomposes above −30 °C to elemental sulfur and iodine.
Samarium bromide may refer to:
Europium bromide may refer to:
This page is based on this
Wikipedia article Text is available under the
CC BY-SA 4.0 license; additional terms may apply.
Images, videos and audio are available under their respective licenses.







