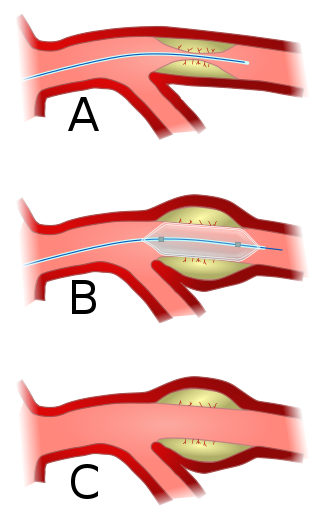
Angioplasty, also known as balloon angioplasty and percutaneous transluminal angioplasty (PTA), is a minimally invasive endovascular procedure used to widen narrowed or obstructed arteries or veins, typically to treat arterial atherosclerosis.

Angiography or arteriography is a medical imaging technique used to visualize the inside, or lumen, of blood vessels and organs of the body, with particular interest in the arteries, veins, and the heart chambers. Modern angiography is performed by injecting a radio-opaque contrast agent into the blood vessel and imaging using X-ray based techniques such as fluoroscopy.
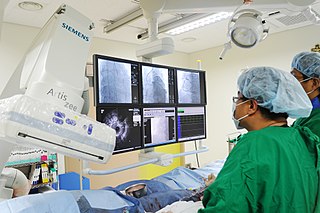
A coronary catheterization is a minimally invasive procedure to access the coronary circulation and blood filled chambers of the heart using a catheter. It is performed for both diagnostic and interventional (treatment) purposes.

Restenosis is the recurrence of stenosis, a narrowing of a blood vessel, leading to restricted blood flow. Restenosis usually pertains to an artery or other large blood vessel that has become narrowed, received treatment to clear the blockage, and subsequently become re-narrowed. This is usually restenosis of an artery, or other blood vessel, or possibly a vessel within an organ.
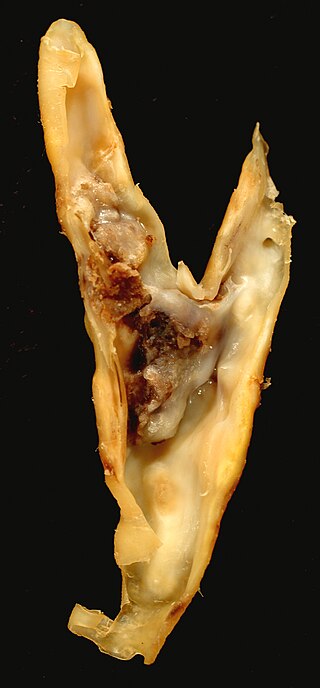
An atheroma, or atheromatous plaque, is an abnormal accumulation of material in the inner layer of an arterial wall.
A vulnerable plaque is a kind of atheromatous plaque – a collection of white blood cells and lipids in the wall of an artery – that is particularly unstable and prone to produce sudden major problems such as a heart attack or stroke.

Cardiac catheterization is the insertion of a catheter into a chamber or vessel of the heart. This is done both for diagnostic and interventional purposes.

Percutaneous coronary intervention (PCI) is a minimally invasive non-surgical procedure used to treat narrowing of the coronary arteries of the heart found in coronary artery disease. The procedure is used to place and deploy coronary stents, a permanent wire-meshed tube, to open narrowed coronary arteries. PCI is considered 'non-surgical' as it uses a small hole in a peripheral artery (leg/arm) to gain access to the arterial system, an equivalent surgical procedure would involve the opening of the chest wall to gain access to the heart area. The term 'coronary angioplasty with stent' is synonymous with PCI. The procedure visualises the blood vessels via fluoroscopic imaging and contrast dyes. PCI is performed by an interventional cardiologists in a catheterization laboratory setting.
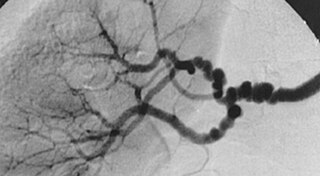
Fibromuscular dysplasia (FMD) is a non-atherosclerotic, non-inflammatory disease of the blood vessels that causes abnormal growth within the wall of an artery. FMD has been found in nearly every arterial bed in the body, although the most commonly affected are the renal and carotid arteries.

A drug-eluting stent (DES) is a tube made of a mesh-like material used to treat narrowed arteries in medical procedures both mechanically and pharmacologically. A DES is inserted into a narrowed artery using a delivery catheter usually inserted through a larger artery in the groin or wrist. The stent assembly has the DES mechanism attached towards the front of the stent, and usually is composed of the collapsed stent over a collapsed polymeric balloon mechanism, the balloon mechanism is inflated and used to expand the meshed stent once in position. The stent expands, embedding into the occluded artery wall, keeping the artery open, thereby improving blood flow. The mesh design allows for stent expansion and also for new healthy vessel endothelial cells to grow through and around it, securing it in place.
Coronary artery anomalies are variations of the coronary circulation, affecting <1% of the general population. Symptoms include chest pain, shortness of breath and syncope, although cardiac arrest may be the first clinical presentation. Several varieties are identified, with a different potential to cause sudden cardiac death.
The history of invasive and interventional cardiology is complex, with multiple groups working independently on similar technologies. Invasive and interventional cardiology is currently closely associated with cardiologists, though the development and most of its early research and procedures were performed by diagnostic and interventional radiologists.
Fractional flow reserve (FFR) is a diagnostic technique used in coronary catheterization. FFR measures pressure differences across a coronary artery stenosis to determine the likelihood that the stenosis impedes oxygen delivery to the heart muscle.

A coronary stent is a tube-shaped device placed in the coronary arteries that supply blood to the heart, to keep the arteries open in patients suffering from coronary heart disease. The vast majority of stents used in modern interventional cardiology are drug-eluting stents (DES). They are used in a medical procedure called percutaneous coronary intervention (PCI). Coronary stents are divided into two broad types: drug-eluting and bare metal stents. As of 2023, drug-eluting stents were used in more than 90% of all PCI procedures. Stents reduce angina and have been shown to improve survival and decrease adverse events after a patient has suffered a heart attack—medically termed an acute myocardial infarction.

Spontaneous coronary artery dissection (SCAD) is an uncommon but potentially lethal condition in which one of the coronary arteries that supply the heart, spontaneously develops a blood collection, or hematoma, within the artery wall due to a tear in the wall. SCAD is one of the arterial dissections that can occur.

A hybrid cardiac surgical procedure in a narrow sense is defined as a procedure that combines a conventional, more invasive surgical part with an interventional part, using some sort of catheter-based procedure guided by fluoroscopy imaging in a hybrid operating room (OR) without interruption. The hybrid technique has a reduced risk of surgical complications and has shown decreased recovery time. It can be used to treat numerous heart diseases and conditions and with the increasing complexity of each case, the hybrid surgical technique is becoming more common.

Endoscopic optical coherence tomography, also intravascular optical coherence tomography is a catheter-based imaging application of optical coherence tomography (OCT). It is capable of acquiring high-resolution images from inside a blood vessel using optical fibers and laser technology.
Intravascular imaging is a catheter based system that allows physicians such as interventional cardiologists to acquire images of diseased vessels from inside the artery. Intravascular imaging provides detailed and accurate measurements of vessel lumen morphology, vessel size, extension of diseased artery segments, vessel size and plaque characteristics. Examples of intravascular imaging modalities are intravascular ultrasound (IVUS) and intracoronary optical coherence tomography.

Intravascular fluorescence is a catheter-based molecular imaging technique that uses near-infrared fluorescence to detect artery wall autofluorescence (NIRAF) or fluorescence generated by molecular agents injected intravenously (NIRF). No commercial systems based on intravascular fluorescence are currently on the market, however, significant steps forwards in intravascular fluorescence imaging technology have been made between 2010-2016. It is typically used to detect functional state of artery wall including some known high-risk features of atherosclerosis. It is usually combined with structural imaging modalities such as Intravascular ultrasound and/or Intracoronary optical coherence tomography, to provide functional information in a morphological context.
Blood vessel disorder generally refers to the narrowing, hardening or enlargement of arteries and veins. It is often due to the build-up of fatty deposits in the lumen of blood vessels or infection of the vessel wall. This can occur in various locations such as coronary blood vessels, peripheral arteries and veins. The narrowed arteries would block the blood supply to different organs and tissues. In severe conditions, it may develop into more critical health problems like myocardial infarction, stroke or heart failure, which are some of the major reasons of death.















