
In chemistry, an ionic crystal is a crystalline form of an ionic compound. They are solids consisting of ions bound together by their electrostatic attraction into a regular lattice. Examples of such crystals are the alkali halides, including potassium fluoride (KF), potassium chloride (KCl), potassium bromide (KBr), potassium iodide (KI), sodium fluoride (NaF). Sodium chloride (NaCl) has a 6:6 co-ordination. The properties of NaCl reflect the strong interactions that exist between the ions. It is a good conductor of electricity when molten, but very poor in the solid state. When fused the mobile ions carry charge through the liquid. They are characterized by strong absorption of infrared radiation and have planes along which they cleave easily. The exact arrangement of ions in an ionic lattice varies according to the size of the ions in the solid.

Potassium bromide (KBr) is a salt, widely used as an anticonvulsant and a sedative in the late 19th and early 20th centuries, with over-the-counter use extending to 1975 in the US. Its action is due to the bromide ion. Potassium bromide is used as a veterinary drug, in antiepileptic medication for dogs.

Potassium permanganate is an inorganic compound with the chemical formula KMnO4. It is a purplish-black crystalline salt, that dissolves in water as K+ and MnO−
4, an intensely pink to purple solution.

Chromate salts contain the chromate anion, CrO2−
4. Dichromate salts contain the dichromate anion, Cr
2O2−
7. They are oxyanions of chromium in the +6 oxidation state and are moderately strong oxidizing agents. In an aqueous solution, chromate and dichromate ions can be interconvertible.

Potassium perchlorate is the inorganic salt with the chemical formula KClO4. Like other perchlorates, this salt is a strong oxidizer when the solid is heated at high temperature although it usually reacts very slowly in solution with reducing agents or organic substances. This colorless crystalline solid is a common oxidizer used in fireworks, ammunition percussion caps, explosive primers, and is used variously in propellants, flash compositions, stars, and sparklers. It has been used as a solid rocket propellant, although in that application it has mostly been replaced by the more performant ammonium perchlorate.

A permanganate is a chemical compound with the manganate(VII) ion, MnO−
4, the conjugate base of permanganic acid. Because the manganese atom has a +7 oxidation state, the permanganate(VII) ion is a strong oxidising agent. The ion is a transition metal ion with a tetrahedral structure. Permanganate solutions are purple in colour and are stable in neutral or slightly alkaline media. The exact chemical reaction depends on the carbon-containing reactants present and the oxidant used. For example, trichloroethane (C2H3Cl3) is oxidised by permanganate ions to form carbon dioxide (CO2), manganese dioxide (MnO2), hydrogen ions (H+), and chloride ions (Cl−).
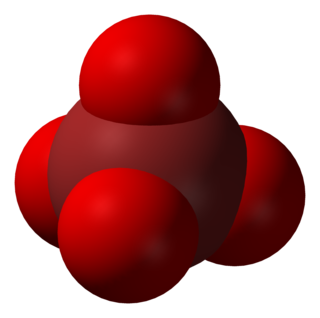
In chemistry, the perbromate ion is the anion having the chemical formula BrO−
4. It is an oxyanion of bromine, the conjugate base of perbromic acid, in which bromine has the oxidation state +7. Unlike its chlorine and iodine analogs, it is difficult to synthesize. It has tetrahedral molecular geometry.
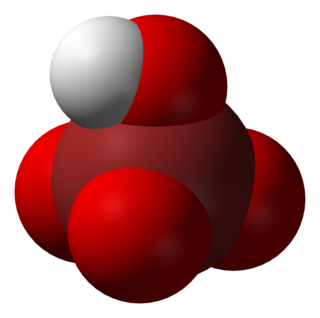
Perbromic acid is the inorganic compound with the formula HBrO4. Perbromic acid is characterized as a colorless liquid which has no characteristic scent. It is an oxoacid of bromine, with an oxidation state of +7. Perbromic acid is a strong acid and strongly oxidizing, though dilute perbromic acid solutions are slow oxidizing agents. It is the most unstable of the halogen(VII) oxoacids. It decomposes rapidly on standing to bromic acid and oxygen, which releases toxic brown bromine vapors. It can be used in the synthesis of perbromate salts, by reacting with a base.
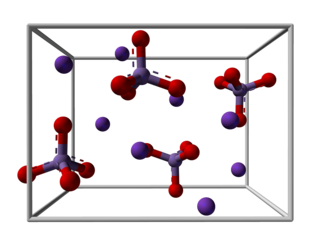
Potassium manganate is the inorganic compound with the formula K2MnO4. This green-colored salt is an intermediate in the industrial synthesis of potassium permanganate, a common chemical. Occasionally, potassium manganate and potassium permanganate are confused, but each compound's properties are distinct.

Potassium chromate is the inorganic compound with the formula K2CrO4. This yellow solid is the potassium salt of the chromate anion. It is a common laboratory chemical, whereas sodium chromate is important industrially.

Bromine can form several different oxides:

Potassium selenate, K
2SeO
4, is an odorless, white solid that forms as the potassium salt of selenic acid.
The periodatonickelates are a series of anions and salts of nickel complexed to the periodate anion. The most important of these salts are the diperiodatonickelates, in which nickel exhibits the +4 oxidation state: these are powerful oxidising agents, capable of oxidising bromate to perbromate.
Sodium perbromate is the chemical compound composed of the sodium ion and the perbromate ion, with the chemical formula NaBrO4.
Barium bromate is a chemical compound composed of the barium ion and the bromate ion, with the chemical formula of Ba(BrO3)2.
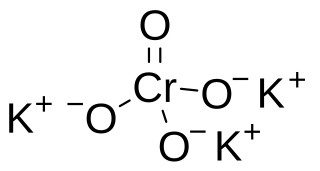
Potassium hypochromate is a chemical compound with the formula K3CrO4 with the unusual Cr5+ ion. This compound is unstable in water but stable in alkaline solution and was found to have a similar crystal structure to potassium hypomanganate.
Perbromyl fluoride is an inorganic compound of bromine, fluorine, and oxygen with the chemical formula BrO3F.

Potassium hexafluoroarsenate is an inorganic chemical compound with the chemical formula KAsF6.

Ammonium perbromate is an inorganic chemical compound with the formula NH4BrO4. It shares similar properties to ammonium perchlorate, but is substantially more difficult to isolate, and has a complex mechanism of decomposition.















