Related Research Articles

Streptococcus is a genus of gram-positive coccus or spherical bacteria that belongs to the family Streptococcaceae, within the order Lactobacillales, in the phylum Bacillota. Cell division in streptococci occurs along a single axis, so as they grow, they tend to form pairs or chains that may appear bent or twisted. This differs from staphylococci, which divide along multiple axes, thereby generating irregular, grape-like clusters of cells. Most streptococci are oxidase-negative and catalase-negative, and many are facultative anaerobes.

Streptococcus pyogenes is a species of Gram-positive, aerotolerant bacteria in the genus Streptococcus. These bacteria are extracellular, and made up of non-motile and non-sporing cocci that tend to link in chains. They are clinically important for humans, as they are an infrequent, but usually pathogenic, part of the skin microbiota that can cause Group A streptococcal infection. S. pyogenes is the predominant species harboring the Lancefield group A antigen, and is often called group A Streptococcus (GAS). However, both Streptococcus dysgalactiae and the Streptococcus anginosus group can possess group A antigen as well. Group A streptococci, when grown on blood agar, typically produce small (2–3 mm) zones of beta-hemolysis, a complete destruction of red blood cells. The name group A (beta-hemolytic) Streptococcus (GABHS) is thus also used.
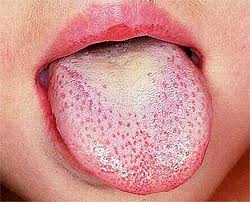
Scarlet fever, also known as scarlatina, is an infectious disease caused by Streptococcus pyogenes, a Group A streptococcus (GAS). The infection is a type of Group A streptococcal infection. It most commonly affects children between five and 15 years of age. The signs and symptoms include a sore throat, fever, headache, swollen lymph nodes, and a characteristic rash. The face is flushed and the rash is red and blanching. It typically feels like sandpaper and the tongue may be red and bumpy. The rash occurs as a result of capillary damage by exotoxins produced by S.pyogenes. On darker-pigmented skin the rash may be hard to discern.

Frederick Griffith (1877–1941) was a British bacteriologist whose focus was the epidemiology and pathology of bacterial pneumonia. In January 1928 he reported what is now known as Griffith's Experiment, the first widely accepted demonstrations of bacterial transformation, whereby a bacterium distinctly changes its form and function.
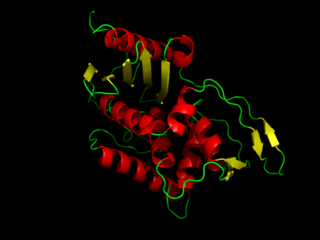
DD-transpeptidase is a bacterial enzyme that catalyzes the transfer of the R-L-αα-D-alanyl moiety of R-L-αα-D-alanyl-D-alanine carbonyl donors to the γ-OH of their active-site serine and from this to a final acceptor. It is involved in bacterial cell wall biosynthesis, namely, the transpeptidation that crosslinks the peptide side chains of peptidoglycan strands.

Lysins, also known as endolysins or murein hydrolases, are hydrolytic enzymes produced by bacteriophages in order to cleave the host's cell wall during the final stage of the lytic cycle. Lysins are highly evolved enzymes that are able to target one of the five bonds in peptidoglycan (murein), the main component of bacterial cell walls, which allows the release of progeny virions from the lysed cell. Cell-wall-containing Archaea are also lysed by specialized pseudomurein-cleaving lysins, while most archaeal viruses employ alternative mechanisms. Similarly, not all bacteriophages synthesize lysins: some small single-stranded DNA and RNA phages produce membrane proteins that activate the host's autolytic mechanisms such as autolysins.
Streptolysins are two hemolytic exotoxins from Streptococcus. Types include streptolysin O, which is oxygen-labile, and streptolysin S, which is oxygen-stable.

Streptococcus dysgalactiae is a gram positive, beta-haemolytic, coccal bacterium belonging to the family Streptococcaceae. It is capable of infecting both humans and animals, but is most frequently encountered as a commensal of the alimentary tract, genital tract, or less commonly, as a part of the skin flora. The clinical manifestations in human disease range from superficial skin-infections and tonsillitis, to severe necrotising fasciitis and bacteraemia. The incidence of invasive disease has been reported to be rising. Several different animal species are susceptible to infection by S. dysgalactiae, but bovine mastitis and infectious arthritis in lambs have been most frequently reported.
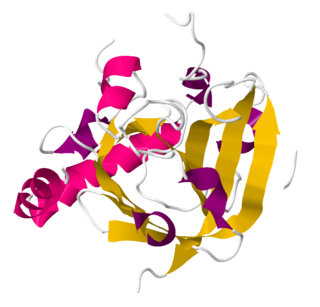
Sortase refers to a group of prokaryotic enzymes that modify surface proteins by recognizing and cleaving a carboxyl-terminal sorting signal. For most substrates of sortase enzymes, the recognition signal consists of the motif LPXTG (Leu-Pro-any-Thr-Gly), then a highly hydrophobic transmembrane sequence, followed by a cluster of basic residues such as arginine. Cleavage occurs between the Thr and Gly, with transient attachment through the Thr residue to the active site Cys residue, followed by transpeptidation that attaches the protein covalently to cell wall components. Sortases occur in almost all Gram-positive bacteria and the occasional Gram-negative bacterium or Archaea, where cell wall LPXTG-mediated decoration has not been reported. Although sortase A, the "housekeeping" sortase, typically acts on many protein targets, other forms of sortase recognize variant forms of the cleavage motif, or catalyze the assembly of pilins into pili.
Perianal cellulitis, also known as perianitis or perianal streptococcal dermatitis, is a bacterial infection affecting the lower layers of the skin (cellulitis) around the anus. It presents as bright redness in the skin and can be accompanied by pain, difficulty defecating, itching, and bleeding. This disease is considered a complicated skin and soft tissue infection (cSSTI) because of the involvement of the deeper soft tissues.

Streptococcal pyrogenic exotoxins also known as erythrogenic toxins, are exotoxins secreted by strains of the bacterial species Streptococcus pyogenes. SpeA and speC are superantigens, which induce inflammation by nonspecifically activating T cells and stimulating the production of inflammatory cytokines. SpeB, the most abundant streptococcal extracellular protein, is a cysteine protease. Pyrogenic exotoxins are implicated as the causative agent of scarlet fever and streptococcal toxic shock syndrome. There is no consensus on the exact number of pyrogenic exotoxins. Serotypes A-C are the most extensively studied and recognized by all sources, but others note up to thirteen distinct types, categorizing speF through speM as additional superantigens. Erythrogenic toxins are known to damage the plasma membranes of blood capillaries under the skin and produce a red skin rash. Past studies have shown that multiple variants of erythrogenic toxins may be produced, depending on the strain of S. pyogenes in question. Some strains may not produce a detectable toxin at all. Bacteriophage T12 infection of S. pyogenes enables the production of speA, and increases virulence.
Bacteriophage T12 is a bacteriophage that infects Streptococcus pyogenes bacteria. It is a proposed species of the family Siphoviridae in the order Caudovirales also known as tailed viruses. It converts a harmless strain of bacteria into a virulent strain. It carries the speA gene which codes for erythrogenic toxin A. speA is also known as streptococcal pyogenic exotoxin A, scarlet fever toxin A, or even scarlatinal toxin. Note that the name of the gene "speA" is italicized; the name of the toxin "speA" is not italicized. Erythrogenic toxin A converts a harmless, non-virulent strain of Streptococcus pyogenes to a virulent strain through lysogeny, a life cycle which is characterized by the ability of the genome to become a part of the host cell and be stably maintained there for generations. Phages with a lysogenic life cycle are also called temperate phages. Bacteriophage T12, proposed member of family Siphoviridae including related speA-carrying bacteriophages, is also a prototypic phage for all the speA-carrying phages of Streptococcus pyogenes, meaning that its genome is the prototype for the genomes of all such phages of S. pyogenes. It is the main suspect as the cause of scarlet fever, an infectious disease that affects small children.
The alpha-D-phosphohexomutases are a large superfamily of enzymes, with members in all three domains of life. Enzymes from this superfamily are ubiquitous in organisms from E. Coli to humans, and catalyze a phosphoryl transfer reaction on a phosphosugar substrate. Four well studied subgroups in the superfamily are:
- Phosphoglucomutase (PGM)
- Phosphoglucomutase/Phosphomannomutase (PGM/PMM)
- Phosphoglucosamine mutase (PNGM)
- Phosphoaceytlglucosamine mutase (PAGM)

Peptidase Do is an enzyme. This enzyme catalyses the following chemical reaction
SpoIVB peptidase is an enzyme. This enzyme catalyses the following chemical reaction
Streptopain is an enzyme. This enzyme catalyses the following chemical reaction
Sortase A is an enzyme. This enzyme catalyses a cell wall sorting reaction, in which a surface protein with a sorting signal containing a LPXTG motif, is cleaved between the Thr and Gly residue.
LPXTGase refers to an endopeptidase enzyme from Streptococci and Staphylococci with the capacity to cleave the carboxy-terminal LPXTG anchor motif of surface proteins similar to Sortase. However, LPXTGase differs significantly from Sortase in several ways: a) it is glycosylated, b) it contains unconventional amino acids, and c) it contains D-amino acids. The latter two characteristics indicate that ribosomes are not involve in the synthesis of LPXTGase. Data suggest that the enzymes responsible for cell wall assembly also assemble LPXTGase.

Glutamic proteases are a group of proteolytic enzymes containing a glutamic acid residue within the active site. This type of protease was first described in 2004 and became the sixth catalytic type of protease. Members of this group of protease had been previously assumed to be an aspartate protease, but structural determination showed it to belong to a novel protease family. The first structure of this group of protease was scytalidoglutamic peptidase, the active site of which contains a catalytic dyad, glutamic acid (E) and glutamine (Q), which give rise to the name eqolisin. This group of proteases are found primarily in pathogenic fungi affecting plant and human.
RopB transcriptional regulator, also known as RopB/Rgg transcriptional regulator is a transcriptional regulator protein that regulates expression of the extracellularly secreted cysteine protease streptococcal pyrogenic exotoxin B (speB) [See Also: erythrogenic toxins] which is an important virulence factor of Streptococcus pyogenes and is responsible for the dissemination of a host of infectious diseases including strep throat, impetigo, streptococcal toxic shock syndrome, necrotizing fasciitis, and scarlet fever. Functional studies suggest that the ropB multigene regulon is responsible for not only global regulation of virulence but also a wide range of functions from stress response, metabolic function, and two-component signaling. Structural studies implicate ropB's regulatory action being reliant on a complex interaction involving quorum sensing with the leaderless peptide signal speB-inducing peptide (SIP) acting in conjunction with a pH sensitive histidine switch.
References
- ↑ Wexler DE, Chenoweth DE, Cleary PP (December 1985). "Mechanism of action of the group A streptococcal C5a inactivator". Proceedings of the National Academy of Sciences of the United States of America. 82 (23): 8144–8. doi: 10.1073/pnas.82.23.8144 . PMC 391459 . PMID 3906656.
- ↑ Bohnsack JF, Mollison KW, Buko AM, Ashworth JC, Hill HR (February 1991). "Group B streptococci inactivate complement component C5a by enzymic cleavage at the C-terminus". The Biochemical Journal. 273 ( Pt 3) (3): 635–40. doi:10.1042/bj2730635. PMC 1149811 . PMID 1996961.
- ↑ Cleary PP, Prahbu U, Dale JB, Wexler DE, Handley J (December 1992). "Streptococcal C5a peptidase is a highly specific endopeptidase". Infection and Immunity. 60 (12): 5219–23. doi:10.1128/iai.60.12.5219-5223.1992. PMC 258300 . PMID 1452354.
- ↑ Anderson ET, Wetherell MG, Winter LA, Olmsted SB, Cleary PP, Matsuka YV (October 2002). "Processing, stability, and kinetic parameters of C5a peptidase from Streptococcus pyogenes". European Journal of Biochemistry. 269 (19): 4839–51. doi: 10.1046/j.1432-1033.2002.03183.x . PMID 12354115.
- ↑ Stafslien DK, Cleary PP (June 2000). "Characterization of the streptococcal C5a peptidase using a C5a-green fluorescent protein fusion protein substrate". Journal of Bacteriology. 182 (11): 3254–8. doi:10.1128/jb.182.11.3254-3258.2000. PMC 94514 . PMID 10809707.
- ↑ Terao Y, Yamaguchi M, Hamada S, Kawabata S (May 2006). "Multifunctional glyceraldehyde-3-phosphate dehydrogenase of Streptococcus pyogenes is essential for evasion from neutrophils". The Journal of Biological Chemistry. 281 (20): 14215–23. doi: 10.1074/jbc.M513408200 . PMID 16565520.