
Phospholipids are a class of lipids whose molecule has a hydrophilic "head" containing a phosphate group and two hydrophobic "tails" derived from fatty acids, joined by an alcohol residue. Marine phospholipids typically have omega-3 fatty acids EPA and DHA integrated as part of the phospholipid molecule. The phosphate group can be modified with simple organic molecules such as choline, ethanolamine or serine.
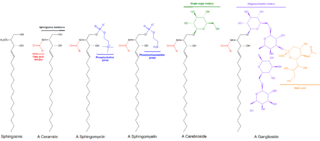
Sphingomyelin is a type of sphingolipid found in animal cell membranes, especially in the membranous myelin sheath that surrounds some nerve cell axons. It usually consists of phosphocholine and ceramide, or a phosphoethanolamine head group; therefore, sphingomyelins can also be classified as sphingophospholipids. In humans, SPH represents ~85% of all sphingolipids, and typically make up 10–20 mol % of plasma membrane lipids.
Sphingomyelin phosphodiesterase D (EC 3.1.4.41, sphingomyelinase D) is an enzyme of the sphingomyelin phosphodiesterase family with systematic name sphingomyelin ceramide-phosphohydrolase. These enzymes catalyse the hydrolysis of sphingomyelin, resulting in the formation of ceramide 1-phosphate and choline:
Phosphorylcholine is the hydrophilic polar head group of some phospholipids, which is composed of a negatively charged phosphate bonded to a small, positively charged choline group. Phosphorylcholine is part of the platelet-activating factor; the phospholipid phosphatidylcholine and sphingomyelin, the only phospholipid of the membrane that is not built with a glycerol backbone. Treatment of cell membranes, like those of RBCs, by certain enzymes, like some phospholipase A2, renders the phosphorylcholine moiety exposed to the external aqueous phase, and thus accessible for recognition by the immune system. Antibodies against phosphorylcholine are naturally occurring autoantibodies that are created by CD5+/B-1 B cells and are referred to as non-pathogenic autoantibodies.
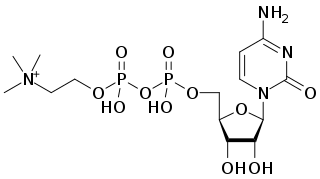
Citicoline (INN), also known as cytidine diphosphate-choline (CDP-choline) or cytidine 5'-diphosphocholine is an intermediate in the generation of phosphatidylcholine from choline, a common biochemical process in cell membranes. Citicoline is naturally occurring in the cells of human and animal tissue, in particular the organs.
In enzymology, sphingosine N-acyltransferases (ceramide synthases (CerS), EC 2.3.1.24) are enzymes that catalyze the chemical reaction of synthesis of ceramide:
In enzymology, a beta-galactoside alpha-2,6-sialyltransferase is an enzyme that catalyzes the chemical reaction
In enzymology, a ganglioside galactosyltransferase is an enzyme that catalyzes the chemical reaction
In enzymology, a 1-alkenyl-2-acylglycerol choline phosphotransferase is an enzyme that catalyzes the chemical reaction
In enzymology, a CDP-diacylglycerol—glycerol-3-phosphate 3-phosphatidyltransferase is an enzyme that catalyzes the chemical reaction

In enzymology, a CDP-diacylglycerol—inositol 3-phosphatidyltransferase is an enzyme that catalyzes the chemical reaction
In enzymology, a CDP-diacylglycerol—serine O-phosphatidyltransferase is an enzyme that catalyzes the chemical reaction
Choline-phosphate cytidylyltransferase is an enzyme that catalyzes the chemical reaction
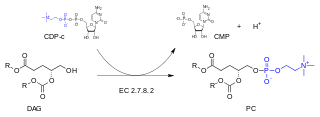
In enzymology, a diacylglycerol cholinephosphotransferase is an enzyme that catalyzes the chemical reaction
In enzymology, an ethanolaminephosphotransferase is an enzyme that catalyzes the chemical reaction
In enzymology, a phosphatidylcholine synthase is an enzyme that catalyzes the chemical reaction
In enzymology, a sphingomyelin synthase is an enzyme that catalyzes the chemical reaction
In enzymology, a sphingosine cholinephosphotransferase is an enzyme that catalyzes the chemical reaction
Acid sphingomyelinase is one of the enzymes that make up the sphingomyelinase (SMase) family, responsible for catalyzing the breakdown of sphingomyelin to ceramide and phosphorylcholine. They are organized into alkaline, neutral, and acidic SMase depending on the pH in which their enzymatic activity is optimal. Acid sphingomyelinases' (aSMases) enzymatic activity can be influenced by drugs, lipids, cations, pH, redox and other proteins in the environment. Specifically aSMases have been shown to have increased enzymatic activity in lysobisphosphatidic acid (LBPA) or phosphatidylinositol (PI) enriched environments, and inhibited activity when phosphorylated derivatives of PI are present.
In enzymology, a ceramide phosphoethanolamine synthase is an enzyme that catalyzes the chemical reaction





