
A cofactor is a non-protein chemical compound or metallic ion that is required for an enzyme's role as a catalyst. Cofactors can be considered "helper molecules" that assist in biochemical transformations. The rates at which these happen are characterized in an area of study called enzyme kinetics. Cofactors typically differ from ligands in that they often derive their function by remaining bound.

Pterin is a heterocyclic compound composed of a pteridine ring system, with a "keto group" and an amino group on positions 4 and 2 respectively. It is structurally related to the parent bicyclic heterocycle called pteridine. Pterins, as a group, are compounds related to pterin with additional substituents. Pterin itself is of no biological significance.

Molybdopterins are a class of cofactors found in most molybdenum-containing and all tungsten-containing enzymes. Synonyms for molybdopterin are: MPT and pyranopterin-dithiolate. The nomenclature for this biomolecule can be confusing: Molybdopterin itself contains no molybdenum; rather, this is the name of the ligand that will bind the active metal. After molybdopterin is eventually complexed with molybdenum, the complete ligand is usually called molybdenum cofactor.

Sulfite oxidase is an enzyme in the mitochondria of all eukaryotes, with exception of the yeasts. It oxidizes sulfite to sulfate and, via cytochrome c, transfers the electrons produced to the electron transport chain, allowing generation of ATP in oxidative phosphorylation. This is the last step in the metabolism of sulfur-containing compounds and the sulfate is excreted.

Lipoyl synthase is an enzyme that belongs to the radical SAM (S-adenosyl methionine) family. Within the radical SAM superfamily, lipoyl synthase is in a sub-family of enzymes that catalyze sulfur insertion reactions. The enzymes in this subfamily differ from general radical SAM enzymes, as they contain two 4Fe-4S clusters. From these clusters, the enzymes obtain the sulfur groups that will be transferred onto the corresponding substrates. This particular enzyme participates in the final step of lipoic acid metabolism, transferring two sulfur atoms from its 4Fe-4S cluster onto the protein N6-(octanoyl)lysine through radical generation. This enzyme is usually localized to the mitochondria. Two organisms that have been extensively studied with regards to this enzyme are Escherichia coli and Mycobacterium tuberculosis. It is currently being studied in other organisms including yeast, plants, and humans.

Molybdenum cofactor biosynthesis protein 1 is a protein that in humans and other animals, fungi, and cellular slime molds, is encoded by the MOCS1 gene.

Molybdenum cofactor synthesis protein 2A and molybdenum cofactor synthesis protein 2B are a pair of proteins that in humans are encoded from the same MOCS2 gene. These two proteins dimerize to form molybdopterin synthase.

Adenylyltransferase and sulfurtransferase MOCS3 is an enzyme that in humans is encoded by the MOCS3 gene.
Molybdopterin synthase (EC 2.8.1.12, MPT synthase) is an enzyme required to synthesize molybdopterin (MPT) from precursor Z (now known as cyclic pyranopterin monophosphate). Molydopterin is subsequently complexed with molybdenum to form molybdenum cofactor (MoCo). MPT synthase catalyses the following chemical reaction:
Radical SAM is a designation for a superfamily of enzymes that use a [4Fe-4S]+ cluster to reductively cleave S-adenosyl-L-methionine (SAM) to generate a radical, usually a 5′-deoxyadenosyl radical (5'-dAdo), as a critical intermediate. These enzymes utilize this radical intermediate to perform diverse transformations, often to functionalize unactivated C-H bonds. Radical SAM enzymes are involved in cofactor biosynthesis, enzyme activation, peptide modification, post-transcriptional and post-translational modifications, metalloprotein cluster formation, tRNA modification, lipid metabolism, biosynthesis of antibiotics and natural products etc. The vast majority of known radical SAM enzymes belong to the radical SAM superfamily, and have a cysteine-rich motif that matches or resembles CxxxCxxC. rSAMs comprise the largest superfamily of metal-containing enzymes.
Sulfur carrier protein ThiS adenylyltransferase is an enzyme with systematic name ATP:(ThiS) adenylyltransferase. This enzyme catalyses the following chemical reaction
Molybdopterin adenylyltransferase is an enzyme with systematic name ATP:molybdopterin adenylyltransferase. This enzyme catalyses the following chemical reaction
Molybdenum cofactor cytidylyltransferase is an enzyme with systematic name CTP:molybdenum cofactor cytidylyltransferase. This enzyme catalyses the following chemical reaction:
Molybdenum cofactor guanylyltransferase is an enzyme with systematic name GTP:molybdenum cofactor guanylyltransferase. This enzyme catalyses the following chemical reaction:
Molybdopterin synthase sulfurtransferase is an enzyme with systematic name persulfurated L-cysteine desulfurase:(molybdopterin-synthase sulfur-carrier protein)-Gly-Gly sulfurtransferase. This enzyme catalyses the following chemical reaction
Molybdopterin molybdotransferase is an enzyme with systematic name adenylyl-molybdopterin:molybdate molybdate transferase (AMP-forming). This enzyme catalyses the following chemical reaction
Cyclic pyranopterin monophosphate synthase is an enzyme with systematic name GTP 8,9-lyase . This enzyme catalyses the following chemical reaction
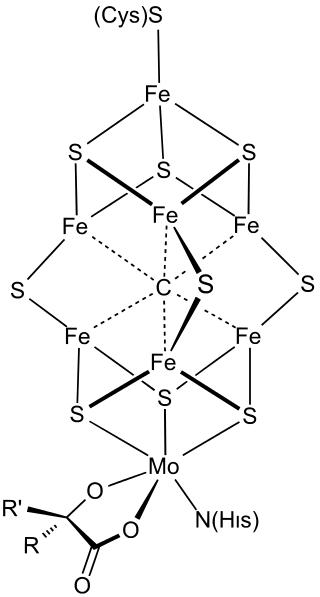
FeMoco (FeMo cofactor) is the primary cofactor of nitrogenase. Nitrogenase is the enzyme that catalyzes the conversion of atmospheric nitrogen molecules N2 into ammonia (NH3) through the process known as nitrogen fixation. Studying FeMoco's role in the reaction mechanism for nitrogen fixation is a potential use case for quantum computers. Even limited quantum computers could enable better simulations of the reaction mechanism.

In enzymology, an aldehyde ferredoxin oxidoreductase (EC 1.2.7.5) is an enzyme that catalyzes the chemical reaction
Molybdenum is an essential element in most organisms. It is most notably present in nitrogenase which is an essential part of nitrogen fixation.











