 | |
 | |
| Names | |
|---|---|
| IUPAC name sodium cyclo-hexaphosphate | |
| Other names Calgon S Glassy sodium Contents | |
| Identifiers | |
| |
3D model (JSmol) | |
| ChemSpider | |
| ECHA InfoCard | 100.030.299 |
| EC Number |
|
| MeSH | sodium+polymetaphosphate |
PubChem CID | |
| UNII | |
CompTox Dashboard (EPA) | |
| |
| |
| Properties | |
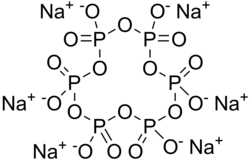
| Na6[(PO3)6] | |
| Molar mass | 611.7704 g mol−1 |
| Appearance | White crystals |
| Odor | odorless |
| Density | 2.484 g/cm3 |
| Melting point | 628 °C (1,162 °F; 901 K) |
| Boiling point | 1,500 °C (2,730 °F; 1,770 K) |
| soluble | |
| Solubility | insoluble in organic solvents |
Refractive index (nD) | 1.482 |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards | Irritant |
| GHS labelling: [2] | |
 | |
| Warning | |
| H319 | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose) | 3.053 g kg−1 |
| Safety data sheet (SDS) | hazard.com |
| Related compounds | |
Other anions | Trisodium phosphate Tetrasodium pyrophosphate Pentasodium triphosphate |
Related compounds | Sodium trimetaphosphate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
Sodium hexametaphosphate (SHMP) is a salt of composition Na6[(PO3)6]. [3] Sodium hexametaphosphate of commerce is typically a mixture of metaphosphates (empirical formula: NaPO3), of which the hexamer is one, and is usually the compound referred to by this name. Such a mixture is more correctly termed sodium polymetaphosphate. They are white solids that dissolve in water.