A protein phosphatase is a phosphatase enzyme that removes a phosphate group from the phosphorylated amino acid residue of its substrate protein. Protein phosphorylation is one of the most common forms of reversible protein posttranslational modification (PTM), with up to 30% of all proteins being phosphorylated at any given time. Protein kinases (PKs) are the effectors of phosphorylation and catalyse the transfer of a γ-phosphate from ATP to specific amino acids on proteins. Several hundred PKs exist in mammals and are classified into distinct super-families. Proteins are phosphorylated predominantly on Ser, Thr and Tyr residues, which account for 79.3, 16.9 and 3.8% respectively of the phosphoproteome, at least in mammals. In contrast, protein phosphatases (PPs) are the primary effectors of dephosphorylation and can be grouped into three main classes based on sequence, structure and catalytic function. The largest class of PPs is the phosphoprotein phosphatase (PPP) family comprising PP1, PP2A, PP2B, PP4, PP5, PP6 and PP7, and the protein phosphatase Mg2+- or Mn2+-dependent (PPM) family, composed primarily of PP2C. The protein Tyr phosphatase (PTP) super-family forms the second group, and the aspartate-based protein phosphatases the third. The protein pseudophosphatases form part of the larger phosphatase family, and in most cases are thought to be catalytically inert, instead functioning as phosphate-binding proteins, integrators of signalling or subcellular traps. Examples of membrane-spanning protein phosphatases containing both active (phosphatase) and inactive (pseudophosphatase) domains linked in tandem are known, conceptually similar to the kinase and pseudokinase domain polypeptide structure of the JAK pseudokinases. A complete comparative analysis of human phosphatases and pseudophosphatases has been completed by Manning and colleagues, forming a companion piece to the ground-breaking analysis of the human kinome, which encodes the complete set of ~536 human protein kinases.

In biochemistry, phosphorylation is the attachment of a phosphate group to a molecule or an ion. This process and its inverse, dephosphorylation, are common in biology. Protein phosphorylation often activates many enzymes.

Lysine (symbol Lys or K) is an α-amino acid that is a precursor to many proteins. It contains an α-amino group (which is in the protonated −NH+
3 form under biological conditions), an α-carboxylic acid group (which is in the deprotonated −COO− form under biological conditions), and a side chain lysyl ((CH2)4NH2), classifying it as a basic, charged (at physiological pH), aliphatic amino acid. It is encoded by the codons AAA and AAG. Like almost all other amino acids, the α-carbon is chiral and lysine may refer to either enantiomer or a racemic mixture of both. For the purpose of this article, lysine will refer to the biologically active enantiomer L-lysine, where the α-carbon is in the S configuration.
Gluconeogenesis (GNG) is a metabolic pathway that results in the generation of glucose from certain non-carbohydrate carbon substrates. It is a ubiquitous process, present in plants, animals, fungi, bacteria, and other microorganisms. In vertebrates, gluconeogenesis occurs mainly in the liver and, to a lesser extent, in the cortex of the kidneys. It is one of two primary mechanisms – the other being degradation of glycogen (glycogenolysis) – used by humans and many other animals to maintain blood sugar levels, avoiding low levels (hypoglycemia). In ruminants, because dietary carbohydrates tend to be metabolized by rumen organisms, gluconeogenesis occurs regardless of fasting, low-carbohydrate diets, exercise, etc. In many other animals, the process occurs during periods of fasting, starvation, low-carbohydrate diets, or intense exercise.
5' AMP-activated protein kinase or AMPK or 5' adenosine monophosphate-activated protein kinase is an enzyme that plays a role in cellular energy homeostasis, largely to activate glucose and fatty acid uptake and oxidation when cellular energy is low. It belongs to a highly conserved eukaryotic protein family and its orthologues are SNF1 in yeast, and SnRK1 in plants. It consists of three proteins (subunits) that together make a functional enzyme, conserved from yeast to humans. It is expressed in a number of tissues, including the liver, brain, and skeletal muscle. In response to binding AMP and ADP, the net effect of AMPK activation is stimulation of hepatic fatty acid oxidation, ketogenesis, stimulation of skeletal muscle fatty acid oxidation and glucose uptake, inhibition of cholesterol synthesis, lipogenesis, and triglyceride synthesis, inhibition of adipocyte lipogenesis, inhibition of adipocyte lipolysis, and modulation of insulin secretion by pancreatic β-cells.
In cell biology, Protein kinase C, commonly abbreviated to PKC (EC 2.7.11.13), is a family of protein kinase enzymes that are involved in controlling the function of other proteins through the phosphorylation of hydroxyl groups of serine and threonine amino acid residues on these proteins, or a member of this family. PKC enzymes in turn are activated by signals such as increases in the concentration of diacylglycerol (DAG) or calcium ions (Ca2+). Hence PKC enzymes play important roles in several signal transduction cascades.
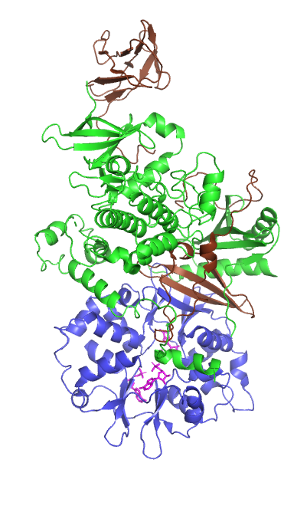
Fructokinase, also known as D-fructokinase or D-fructose (D-mannose) kinase, is an enzyme of the liver, intestine, and kidney cortex. Fructokinase is in a family of enzymes called transferases, meaning that this enzyme transfers functional groups; it is also considered a phosphotransferase since it specifically transfers a phosphate group. Fructokinase specifically catalyzes the transfer of a phosphate group from adenosine triphosphate to fructose as the initial step in its utilization. The main role of fructokinase is in carbohydrate metabolism, more specifically, sucrose and fructose metabolism. The reaction equation is as follows:
Carnosine synthase is an enzyme that catalyzes the chemical reaction
In enzymology, arginine kinase is an enzyme that catalyzes the chemical reaction

Phosphoserine phosphatase is an enzyme that in humans is encoded by the PSPH gene.

Fructosamine-3-kinase is an enzyme that in humans is encoded by the FN3K gene.

Ketosamine-3-kinase is an enzyme that in humans is encoded by the FN3KRP (fructosamine-3-kinase-related-protein) gene.
The Walker A and Walker B motifs are protein sequence motifs, known to have highly conserved three-dimensional structures. These were first reported in ATP-binding proteins by Walker and co-workers in 1982.

3-Deoxyglucosone (3DG) is a sugar that is notable because it is a marker for diabetes. 3DG reacts with protein to form advanced glycation end-products (AGEs), which contribute to diseases such as the vascular complications of diabetes, atherosclerosis, hypertension, Alzheimer's disease, inflammation, and aging.
In molecular biology the fructosamine kinase family is a family of enzymes. This family includes eukaryotic fructosamine-3-kinase enzymes which may initiate a process leading to the deglycation of fructoselysine and of glycated proteins and in the phosphorylation of 1-deoxy-1-morpholinofructose, fructoselysine, fructoseglycine, fructose and glycated lysozyme. The family also includes ketosamine-3-kinases (KT3K). Ketosamines derive from a non-enzymatic reaction between a sugar and a protein. Ketosamine-3-kinases (KT3K) catalyse the phosphorylation of the ketosamine moiety of glycated proteins. The instability of a phosphorylated ketosamine leads to its degradation, and KT3K is thus thought to be involved in protein repair.
Protein-fructosamine 3-kinase (EC 2.7.1.171, FN3K, fructosamine 3-kinase) is an enzyme with systematic name ATP:(protein)-N6-D-fructosyl-L-lysine 3-phosphotransferase. This enzyme catalyses the following chemical reaction
5-Phosphonooxy-L-lysine phospho-lyase (EC 4.2.3.134, 5-phosphohydroxy-L-lysine ammoniophospholyase, AGXT2L2 (gene)) is an enzyme with systematic name (5R)-5-phosphonooxy-L-lysine phosphate-lyase (deaminating; (S)-2-amino-6-oxohexanoate-forming). This enzyme catalyses the following chemical reaction
Fructoselysine is an Amadori adduct of glucose to lysine.
Fructoselysine-6-kinase is a kinase enzyme.
Pseudokinases are catalytically-deficient pseudoenzyme variants of protein kinases that are represented in all kinomes across the kingdoms of life. Pseudokinases have both physiological and pathophysiological functions.







