In chemistry, water(s) of crystallization or water(s) of hydration are water molecules that are present inside crystals. Water is often incorporated in the formation of crystals from aqueous solutions. In some contexts, water of crystallization is the total mass of water in a substance at a given temperature and is mostly present in a definite (stoichiometric) ratio. Classically, "water of crystallization" refers to water that is found in the crystalline framework of a metal complex or a salt, which is not directly bonded to the metal cation.

Manganese(II) chloride is the dichloride salt of manganese, MnCl2. This inorganic chemical exists in the anhydrous form, as well as the dihydrate (MnCl2·2H2O) and tetrahydrate (MnCl2·4H2O), with the tetrahydrate being the most common form. Like many Mn(II) species, these salts are pink, with the paleness of the color being characteristic of transition metal complexes with high spin d5 configurations.

Iron(II) acetate is a coordination complex with formula Fe(CH3COO)2. It is a white solid, although impure samples can be slightly colored. A light green tetrahydrate is also known, which is highly soluble in water.
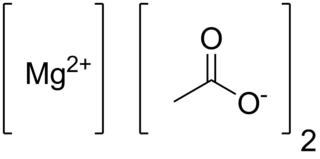
Anhydrous magnesium acetate has the chemical formula Mg(C2H3O2)2 and in its hydrated form, magnesium acetate tetrahydrate, it has the chemical formula Mg(CH3COO)2 • 4H2O. In this compound magnesium has an oxidation state of 2+. Magnesium acetate is the magnesium salt of acetic acid. It is deliquescent and upon heating, it decomposes to form magnesium oxide. Magnesium acetate is commonly used as a source of magnesium in biological reactions.

Tris(acetylacetonato) iron(III), often abbreviated Fe(acac)3, is a ferric coordination complex featuring acetylacetonate (acac) ligands, making it one of a family of metal acetylacetonates. It is a red air-stable solid that dissolves in nonpolar organic solvents.

Nickel(II) acetate is the name for the coordination compounds with the formula Ni(CH3CO2)2·x H2O where x can be 0, 2, and 4. The green tetrahydrate Ni(CH3CO2)2·4 H2O is most common. It is used for electroplating.

Dysprosium acetylacetonate is a chemical compound of dysprosium with formula Dy(C5H7O2)3(H2O)n.
Thullium(III) fluoride is an inorganic compound with the chemical formula TmF3.

Neodymium acetate is an inorganic salt composed of a neodymium atom trication and three acetate groups as anions where neodymium exhibits the +3 oxidation state. It has a chemical formula of Nd(CH3COO)3 although it can be informally referred to as NdAc because Ac is an informal symbol for acetate. It commonly occurs as a light purple powder.
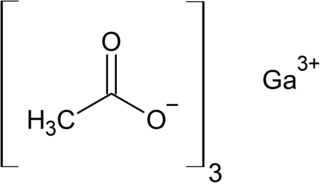
Gallium acetate is a salt composed of a gallium atom trication and three acetate groups as anions where gallium exhibits the +3 oxidation state. It has a chemical formula of Ga(CH3COO)3 although it can be informally referred to as GaAc because Ac is an informal symbol for acetate. Gallium is moderately water-soluble and decomposes to gallium oxide when heated to around 70 °C. Gallium acetate, like other acetate compounds, is a good precursor to ultra-pure compounds, catalysts and nanoscale materials. Gallium acetate is being considered as a substitute in de-icing compounds like calcium chloride and magnesium chloride.

Europium(III) acetate is an inorganic salt of europium and acetic acid with the chemical formula of Eu(CH3COO)3. In this compound, europium exhibits the +3 oxidation state. It can exist in the anhydrous form, sesquihydrate and tetrahydrate. Its hydrate molecule is a dimer.

Holmium acetate is the acetate salt of holmium, with a chemical formula of Ho(CH3COO)3.
Europium(III) chromate is a chemical compound composed of europium, chromium and oxygen with europium in the +3 oxidation state, chromium in the +5 oxidation state and oxygen in the -2 oxidation state. It has the chemical formula of EuCrO4.

Holmium acetylacetonate is a coordination complex, with the chemical formula of Ho(C5H7O2)3 or Ho(acac)3. It can be obtained via the reaction between metallic holmium or holmium(III) hydride with acetylacetone, or via the reaction between holmium(III) chloride and ammonium acetylacetonate. Its anhydrous form is stable in a dry atmosphere but forms a hydrate in humid air.
Thulium(II) fluoride is one of the fluoride salts of the lanthanide metal thulium, with the chemical compound of TmF2. It can react with zirconium tetrafluoride at 900 °C to form TmZrF6, which has a hexagonal structure. In addition, low-temperature Mössbauer spectroscopy and some theoretical studies of thulium(II) fluoride have also been reported.
Erbium compounds are compounds containing the element erbium (Er). These compounds are usually dominated by erbium in the +3 oxidation state, although the +2, +1 and 0 oxidation states have also been reported.
Scandium acetate is an inorganic compound, with the chemical formula of Sc(CH3COO)3. It exists in the anhydrous and the hydrate forms. It can be obtained by reacting scandium hydroxide with aqueous acetic acid. It is a water-soluble crystal that decomposes into scandium oxide at high temperature. It can be used to prepare other scandium-containing materials.

Terbium(III) acetate is the acetate salt of terbium, with a chemical formula of Tb(CH3COO)3.

Gadolinium acetate is the acetate salt of the lanthanide element gadolinium, with the chemical formula Gd(CH3COO)3. It is a colorless crystal that is soluble in water and can form a hydrate. Its tetrahydrate has ground state ferromagnetism.

Lanthanum acetylacetonate is a coordination compound with the chemical formula La(C5H7O2)3, or abbreviated as La(acac)3. Its instability constants (logYn) are 3.65, 5.13 and 6.12 (corresponding to n=1, 2, 3). It can be prepared by the reaction of lanthanum alkoxide and acetylacetone. Its tetrahydrate decomposes into monohydrate at 110 °C, obtains the anhydrous form at 150 °C, undergoes La(CH3COO)(acac)2 and La(CH3COO)2(acac), and at 180~285 °C lanthanum acetate is produced. It can be used to prepare NaLaS2, La2Zr2O7 and other materials.















