
Phosphoglucomutase is an enzyme that transfers a phosphate group on an α-D-glucose monomer from the 1 to the 6 position in the forward direction or the 6 to the 1 position in the reverse direction.
In organic chemistry, a tetrose is a monosaccharide with 4 carbon atoms. They have either an aldehyde functional group in position 1 (aldotetroses) or a ketone group in position 2 (ketotetroses).

Glycogen phosphorylase is one of the phosphorylase enzymes. Glycogen phosphorylase catalyzes the rate-limiting step in glycogenolysis in animals by releasing glucose-1-phosphate from the terminal alpha-1,4-glycosidic bond. Glycogen phosphorylase is also studied as a model protein regulated by both reversible phosphorylation and allosteric effects.
In biochemistry, dephosphorylation is the removal of a phosphate group from an organic compound by hydrolysis. It is a reversible post-translational modification. Dephosphorylation and its counterpart, phosphorylation, activate and deactivate enzymes by detaching or attaching phosphoric esters and anhydrides. A notable occurrence of dephosphorylation is the conversion of ATP to ADP and inorganic phosphate.
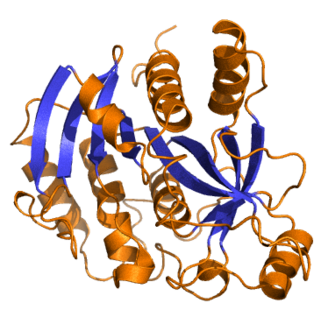
Purine nucleoside phosphorylase, PNP, PNPase or inosine phosphorylase is an enzyme that in humans is encoded by the NP gene. It catalyzes the chemical reaction

Myophosphorylase or glycogen phosphorylase, muscle associated (PYGM) is the muscle isoform of the enzyme glycogen phosphorylase and is encoded by the PYGM gene. This enzyme helps break down glycogen into glucose-1-phosphate, so it can be used within the muscle cell. Mutations in this gene are associated with McArdle disease, a glycogen storage disease of muscle.

Sucrose phosphorylase is an important enzyme in the metabolism of sucrose and regulation of other metabolic intermediates. Sucrose phosphorylase is in the class of hexosyltransferases. More specifically it has been placed in the retaining glycoside hydrolases family although it catalyzes a transglycosidation rather than hydrolysis. Sucrose phosphorylase catalyzes the conversion of sucrose to D-fructose and α-D-glucose-1-phosphate. It has been shown in multiple experiments that the enzyme catalyzes this conversion by a double displacement mechanism.
In enzymology, a quinoprotein glucose dehydrogenase is an enzyme that catalyzes the chemical reaction
In enzymology, a phosphopentomutase is an enzyme that catalyzes the chemical reaction
The enzyme 6-phospho-β-glucosidase (EC 3.2.1.86) catalyzes the following reaction:
In enzymology, an alpha,alpha-trehalose-phosphate synthase (UDP-forming) is an enzyme that catalyzes the chemical reaction

In enzymology, a maltose phosphorylase is an enzyme that catalyzes the chemical reaction
In enzymology, a pyrimidine-nucleoside phosphorylase is an enzyme that catalyzes the chemical reaction

Thymidine phosphorylase is an enzyme that is encoded by the TYMP gene and catalyzes the reaction:
In enzymology, an uridine phosphorylase is an enzyme that catalyzes the chemical reaction

In enzymology, a glucose-1-phosphate adenylyltransferase is an enzyme that catalyzes the chemical reaction
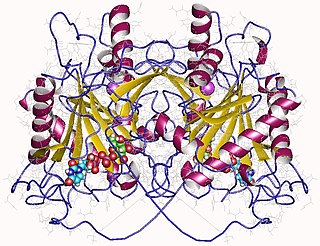
In enzymology, an UDP-glucose—hexose-1-phosphate uridylyltransferase is an enzyme that catalyzes the chemical reaction

In enzymology, an UDP-N-acetylglucosamine diphosphorylase is an enzyme that catalyzes the chemical reaction
Glycogen phosphorylase, liver form (PYGL), also known as human liver glycogen phosphorylase (HLGP), is an enzyme that in humans is encoded by the PYGL gene on chromosome 14. This gene encodes a homodimeric protein that catalyses the cleavage of alpha-1,4-glucosidic bonds to release glucose-1-phosphate from liver glycogen stores. This protein switches from inactive phosphorylase B to active phosphorylase A by phosphorylation of serine residue 14. Activity of this enzyme is further regulated by multiple allosteric effectors and hormonal controls. Humans have three glycogen phosphorylase genes that encode distinct isozymes that are primarily expressed in liver, brain and muscle, respectively. The liver isozyme serves the glycemic demands of the body in general while the brain and muscle isozymes supply just those tissues. In glycogen storage disease type VI, also known as Hers disease, mutations in liver glycogen phosphorylase inhibit the conversion of glycogen to glucose and results in moderate hypoglycemia, mild ketosis, growth retardation and hepatomegaly. Alternative splicing results in multiple transcript variants encoding different isoforms [provided by RefSeq, Feb 2011].
Edith Wilson Miles is a biochemist known for her work on the structure and function of enzymes, especially her work on tryptophan synthase.










