
A protease is an enzyme that catalyzes proteolysis, breaking down proteins into smaller polypeptides or single amino acids, and spurring the formation of new protein products. They do this by cleaving the peptide bonds within proteins by hydrolysis, a reaction where water breaks bonds. Proteases are involved in numerous biological pathways, including digestion of ingested proteins, protein catabolism, and cell signaling.
A metalloproteinase, or metalloprotease, is any protease enzyme whose catalytic mechanism involves a metal. An example is ADAM12 which plays a significant role in the fusion of muscle cells during embryo development, in a process known as myogenesis.

Tosyl phenylalanyl chloromethyl ketone (TPCK) is a protease inhibitor. Its structural formula is 1-chloro-3-tosylamido-4-phenyl-2-butanone.

Papain, also known as papaya proteinase I, is a cysteine protease enzyme present in papaya and mountain papaya. It is the namesake member of the papain-like protease family.

The mountain papaya also known as mountain pawpaw, papayuelo, chamburo, or simply "papaya" is a species of the genus Vasconcellea, native to the Andes of northwestern South America from Colombia south to central Chile, typically growing at altitudes of 1,500–3,000 metres (4,900–9,800 ft).

A catalytic triad is a set of three coordinated amino acids that can be found in the active site of some enzymes. Catalytic triads are most commonly found in hydrolase and transferase enzymes. An acid-base-nucleophile triad is a common motif for generating a nucleophilic residue for covalent catalysis. The residues form a charge-relay network to polarise and activate the nucleophile, which attacks the substrate, forming a covalent intermediate which is then hydrolysed to release the product and regenerate free enzyme. The nucleophile is most commonly a serine or cysteine amino acid, but occasionally threonine or even selenocysteine. The 3D structure of the enzyme brings together the triad residues in a precise orientation, even though they may be far apart in the sequence.
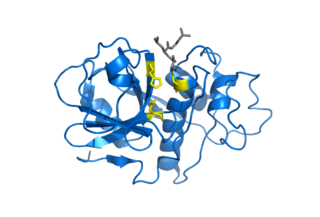
Cysteine proteases, also known as thiol proteases, are hydrolase enzymes that degrade proteins. These proteases share a common catalytic mechanism that involves a nucleophilic cysteine thiol in a catalytic triad or dyad.
Neurophysins are carrier proteins which transport the hormones oxytocin and vasopressin to the posterior pituitary from the paraventricular and supraoptic nucleus of the hypothalamus, respectively. Inside the neurosecretory granules, the analogous neurophysin I and II form stabilizing complexes via covalent interactions. Stabilizing neurophysin-hormone complexes that are formed within neurosecretory granules located in the posterior pituitary gland aid in intra-axonal transport. During intra-axonal transport, the neurophysin's are believed to prevent the bound hormone from leaking into the cytoplasmic space and proteolytic digestion via enzymes. However, due to the low concentration of neurophysin in the blood, it is likely the protein-hormone complex dissociates, indicating the neurophysin does not aid in transporting the hormone through the circulatory system.
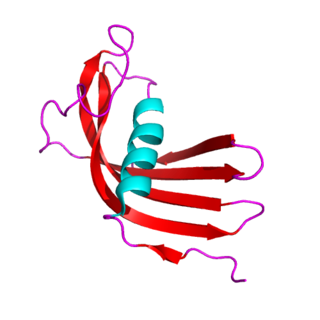
The cystatins are a family of cysteine protease inhibitors which share a sequence homology and a common tertiary structure of an alpha helix lying on top of an anti-parallel beta sheet. The family is subdivided as described below.

Ficain also known as ficin, debricin, or higueroxyl delabarre is a proteolytic enzyme extracted from the latex sap from the stems, leaves, and unripe fruit of the American wild fig tree Ficus insipida.

Actinidain is a type of cysteine protease enzyme found in fruits including kiwifruit, pineapple, mango, banana, figs, and papaya. This enzyme is part of the peptidase C1 family of papain-like proteases.

Ubiquitin carboxyl-terminal hydrolase 20 is an enzyme that in humans is encoded by the USP20 gene.
Proteases are in use, or have been proposed or tried, for a number of purposes related to medicine or surgery. Some preparations involving protease have undergone successful clinical trials and have regulatory authorization; and some further ones have shown apparently useful effects in experimental medical studies. Proteases have also been used by proponents of alternative therapies, or identified in materials of traditional or folk medicine. A serine protease of human origin, activated protein C, was produced in recombinant form and marketed as Drotrecogin alfa and licensed for intensive-care treatment of severe sepsis. It was voluntarily withdrawn by the manufacturer in 2011 after being shown to be ineffective.
Cathepsin X is an enzyme. This enzyme catalyses the following chemical reaction

An Oligopeptidase is an enzyme that cleaves peptides but not proteins. This property is due to its structure: the active site of this enzyme is located at the end of a narrow cavity which can only be reached by peptides.
Glycyl endopeptidase is an enzyme. This enzyme catalyses the following chemical reaction
Caricain is an enzyme. This enzyme catalyses the following chemical reaction: Hydrolysis of proteins with broad specificity for peptide bonds, similar to those of papain and chymopapain

Zingibain, zingipain, or ginger protease is a cysteine protease enzyme found in ginger rhizomes. It catalyses the preferential cleavage of peptides with a proline residue at the P2 position. It has two distinct forms, ginger protease I (GP-I) and ginger protease II (GP-II).
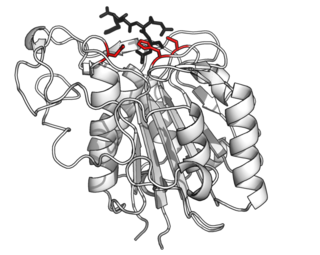
Asparagine endopeptidase is a proteolytic enzyme from C13 peptidase family which hydrolyses a peptide bond using the thiol group of a cysteine residue as a nucleophile. It is also known as asparaginyl endopeptidase, citvac, proteinase B, hemoglobinase, PRSC1 gene product or LGMN, vicilin peptidohydrolase and bean endopeptidase. In humans it is encoded by the LGMN gene.

Papain-like proteases are a large protein family of cysteine protease enzymes that share structural and enzymatic properties with the group's namesake member, papain. They are found in all domains of life. In animals, the group is often known as cysteine cathepsins or, in older literature, lysosomal peptidases. In the MEROPS protease enzyme classification system, papain-like proteases form Clan CA. Papain-like proteases share a common catalytic dyad active site featuring a cysteine amino acid residue that acts as a nucleophile.

















