
The chalcogens are the chemical elements in group 16 of the periodic table. This group is also known as the oxygen family. Group 16 consists of the elements oxygen (O), sulfur (S), selenium (Se), tellurium (Te), and the radioactive elements polonium (Po) and livermorium (Lv). Often, oxygen is treated separately from the other chalcogens, sometimes even excluded from the scope of the term "chalcogen" altogether, due to its very different chemical behavior from sulfur, selenium, tellurium, and polonium. The word "chalcogen" is derived from a combination of the Greek word khalkόs (χαλκός) principally meaning copper, and the Latinized Greek word genēs, meaning born or produced.

Lutetium is a chemical element; it has symbol Lu and atomic number 71. It is a silvery white metal, which resists corrosion in dry air, but not in moist air. Lutetium is the last element in the lanthanide series, and it is traditionally counted among the rare earth elements; it can also be classified as the first element of the 6th-period transition metals.
Chalcogenide glass is a glass containing one or more chalcogens. Up until recently, chalcogenide glasses (ChGs) were believed to be predominantly covalently bonded materials and classified as covalent network solids. A most recent and extremely comprehensive university study of more than 265 different ChG elemental compositions, representing 40 different elemental families now shows that the vast majority of chalcogenide glasses are more accurately defined as being predominantly bonded by the weaker van der Waals forces of atomic physics and more accurately classified as van der Waals network solids. They are not exclusively bonded by these weaker vdW forces, and do exhibit varying percentages of covalency, based upon their specific chemical makeup. Polonium is also a chalcogen but is not used because of its strong radioactivity. Chalcogenide materials behave rather differently from oxides, in particular their lower band gaps contribute to very dissimilar optical and electrical properties.
Organoselenium chemistry is the science exploring the properties and reactivity of organoselenium compounds, chemical compounds containing carbon-to-selenium chemical bonds. Selenium belongs with oxygen and sulfur to the group 16 elements or chalcogens, and similarities in chemistry are to be expected. Organoselenium compounds are found at trace levels in ambient waters, soils and sediments.
Lead selenide (PbSe), or lead(II) selenide, a selenide of lead, is a semiconductor material. It forms cubic crystals of the NaCl structure; it has a direct bandgap of 0.27 eV at room temperature. A grey solid, it is used for manufacture of infrared detectors for thermal imaging. The mineral clausthalite is a naturally occurring lead selenide.

Selenium compounds are compounds containing the element selenium (Se). Among these compounds, selenium has various oxidation states, the most common ones being −2, +4, and +6. Selenium compounds exist in nature in the form of various minerals, such as clausthalite, guanajuatite, tiemannite, crookesite etc., and can also coexist with sulfide minerals such as pyrite and chalcopyrite. For many mammals, selenium compounds are essential. For example, selenomethionine and selenocysteine are selenium-containing amino acids present in the human body. Selenomethionine participates in the synthesis of selenoproteins. The reduction potential and pKa (5.47) of selenocysteine are lower than those of cysteine, making some proteins have antioxidant activity. Selenium compounds have important applications in semiconductors, glass and ceramic industries, medicine, metallurgy and other fields.

Sodium selenide is an inorganic compound of sodium and selenium with the chemical formula Na2Se.

Trioctylphosphine is an organophosphorus compound with the formula P(C8H17)3 sometimes abbreviated TOP. It is usually encountered as a syrup. The compound is colorless.
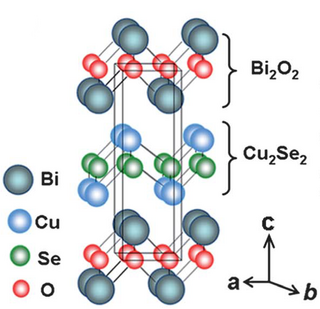
Oxyselenides are a group of chemical compounds that contain oxygen and selenium atoms. Oxyselenides can form a wide range of structures in compounds containing various transition metals, and thus can exhibit a wide range of properties. Most importantly, oxyselenides have a wide range of thermal conductivity, which can be controlled with changes in temperature in order to adjust their thermoelectric performance. Current research on oxyselenides indicates their potential for significant application in electronic materials.
Indium(II) selenide (InSe) is an inorganic compound composed of indium and selenium. It is a III-VI layered semiconductor. The solid has a structure consisting of two-dimensional layers bonded together only by van der Waals forces. Each layer has the atoms in the order Se-In-In-Se.

Lutetium(III) fluoride is an inorganic compound with a chemical formula LuF3.

Lutetium(III) hydroxide is an inorganic compound with the chemical formula Lu(OH)3.

Lutetium(III) acetate is the acetate salt of lutetium with the chemical formula of Lu(CH3COO)3.

Europium compounds are compounds formed by the lanthanide metal europium (Eu). In these compounds, europium generally exhibits the +3 oxidation state, such as EuCl3, Eu(NO3)3 and Eu(CH3COO)3. Compounds with europium in the +2 oxidation state are also known. The +2 ion of europium is the most stable divalent ion of lanthanide metals in aqueous solution. Many europium compounds fluoresce under ultraviolet light due to the excitation of electrons to higher energy levels. Lipophilic europium complexes often feature acetylacetonate-like ligands, e.g., Eufod.

Lutetium(III) iodide or lutetium iodide is an inorganic compound consisting of iodine and lutetium, with the chemical formula of LuI3.
Lutetium compounds are compounds formed by the lanthanide metal lutetium (Lu). In these compounds, lutetium generally exhibits the +3 oxidation state, such as LuCl3, Lu2O3 and Lu2(SO4)3. Aqueous solutions of most lutetium salts are colorless and form white crystalline solids upon drying, with the common exception of the iodide. The soluble salts, such as nitrate, sulfate and acetate form hydrates upon crystallization. The oxide, hydroxide, fluoride, carbonate, phosphate and oxalate are insoluble in water.

Barium selenide is an inorganic compound, with the chemical formula of BaSe. It is a white solid although typically samples are colored owing to the effects of air oxidation.
Thulium(III) selenide is an inorganic compound with the chemical formula Tm2Se3.
Erbium(III) selenide is an inorganic compound with a chemical formula of Er2Se3.
Holmium(III) selenide is an inorganic compound with the chemical formula Ho2Se3.











