
Aconitine is an alkaloid toxin produced by various plant species belonging to the genus Aconitum, commonly known by the names wolfsbane and monkshood. Aconitine is notorious for its toxic properties.
Total synthesis, a specialized area within organic chemistry, focuses on constructing complex organic compounds, especially those found in nature, using laboratory methods. It often involves synthesizing natural products from basic, commercially available starting materials. Total synthesis targets can also be organometallic or inorganic. While total synthesis aims for complete construction from simple starting materials, modifying or partially synthesizing these compounds is known as semisynthesis.

Poison dart frog is the common name of a group of frogs in the family Dendrobatidae which are native to tropical Central and South America. These species are diurnal and often have brightly colored bodies. This bright coloration is correlated with the toxicity of the species, making them aposematic. Some species of the family Dendrobatidae exhibit extremely bright coloration along with high toxicity — a feature derived from their diet of ants, mites and termites— while species which eat a much larger variety of prey have cryptic coloration with minimal to no amount of observed toxicity. Many species of this family are threatened due to human infrastructure encroaching on their habitats.

Batrachotoxin (BTX) is an extremely potent cardiotoxic and neurotoxic steroidal alkaloid found in certain species of beetles, birds, and frogs. The name is from the Greek word βάτραχος, bátrachos, 'frog'. Structurally-related chemical compounds are often referred to collectively as batrachotoxins. In certain frogs, this alkaloid is present mostly on the skin. Such frogs are among those used for poisoning darts. Batrachotoxin binds to and irreversibly opens the sodium channels of nerve cells and prevents them from closing, resulting in paralysis and death. No antidote is known.

Epibatidine is a chlorinated alkaloid that is secreted by the Ecuadoran frog Epipedobates anthonyi and poison dart frogs from the Ameerega genus. It was discovered by John W. Daly in 1974, but its structure was not fully elucidated until 1992. Whether epibatidine occurs naturally remains controversial due to challenges in conclusively identifying the compound from the limited samples collected by Daly. By the time that high-resolution spectrometry was used in 1991, there remained less than one milligram of extract from Daly's samples, raising concerns about possible contamination. Samples from other batches of the same species of frog failed to yield epibatidine.

Lupinine is a quinolizidine alkaloid present in the genus Lupinus of the flowering plant family Fabaceae. The scientific literature contains many reports on the isolation and synthesis of this compound as well as a vast number of studies on its biosynthesis from its natural precursor, lysine. Studies have shown that lupinine hydrochloride is a mildly toxic acetylcholinesterase inhibitor and that lupinine has an inhibitory effect on acetylcholine receptors. The characteristically bitter taste of lupin beans, which come from the seeds of Lupinus plants, is attributable to the quinolizidine alkaloids which they contain, rendering them unsuitable for human and animal consumption unless handled properly. However, because lupin beans have potential nutritional value due to their high protein content, efforts have been made to reduce their alkaloid content through the development of "sweet" varieties of Lupinus.

The strawberry poison frog, strawberry poison-dart frog or blue jeans poison frog is a species of small poison dart frog found in Central America. It is common throughout its range, which extends from eastern central Nicaragua through Costa Rica and northwestern Panama. The species is often found in humid lowlands and premontane forest, but large populations are also found in disturbed areas such as plantations. The strawberry poison frog is perhaps most famous for its widespread variation in coloration, comprising approximately 15–30 color morphs, most of which are presumed to be true-breeding. O. pumilio, while not the most poisonous of the dendrobatids, is the most toxic member of its genus.

The dyeing poison dart frog, also known as the cobalt poison frog, tinc, is a species of poison dart frog. It is among the most variably colored and largest species of poison dart frogs, typically reaching snout–vent lengths of about 50 mm (2.0 in). It is distributed in the eastern portion of the Guiana Shield, including parts of French Guiana, Guyana, Suriname, and Brazil. These types of frogs usually like to stay in the ground as it is easier to catch prey. They tend to also stay in bodies of water like rivers or climb trees. Because of this they mostly inhabit near the Amazon Rainforest. To mate they have special rituals and leave their tadpoles in freshwater.
The Negishi coupling is a widely employed transition metal catalyzed cross-coupling reaction. The reaction couples organic halides or triflates with organozinc compounds, forming carbon-carbon bonds (C-C) in the process. A palladium (0) species is generally utilized as the catalyst, though nickel is sometimes used. A variety of nickel catalysts in either Ni0 or NiII oxidation state can be employed in Negishi cross couplings such as Ni(PPh3)4, Ni(acac)2, Ni(COD)2 etc.

Phyllobates is a genus of poison dart frogs native to Central and South America, from Nicaragua to Colombia. There are 3 different Colombian species of Phyllobates, considered highly toxic species due to the poison they contain in the wild.

Oophaga sylvatica, sometimes known as its Spanish name diablito, is a species of frog in the family Dendrobatidae found in Southwestern Colombia and Northwestern Ecuador. Its natural habitat is lowland and submontane rainforest; it can, however, survive in moderately degraded areas, at least in the more humid parts of its range. It is a very common frog in Colombia, but has disappeared from much of its Ecuadorian range. It is threatened by habitat loss (deforestation) and agricultural pollution and sometimes seen in the international pet trade.

Pumiliotoxin 251D is a toxic organic compound. It is found in the skin of poison frogs from the genera Dendrobates, Epipedobates, Minyobates, and Phyllobates and toads from the genus Melanophryniscus. Its name comes from the pumiliotoxin family (PTXs) and its molecular mass of 251 daltons. When the toxin enters the bloodstream through cuts in the skin or by ingestion, it can cause hyperactivity, convulsions, cardiac arrest and ultimately death. It is especially toxic to arthropods, even at low concentrations.

Pumiliotoxins (PTXs), are one of several toxins found in the skin of poison dart frogs. The frog species, P. bibronii also produces PTXs to deter predators. Closely related, though more toxic, are allopumiliotoxins, (aPTXs). Other toxins found in the skin of poison frogs include decahydroquinolines (DHQs), izidines, coccinellines, and spiropyrrolizidine alkaloids. Pumiliotoxins are very poisonous in high concentrations. Pumiliotoxins are much weaker than batrachotoxins, ranging between 100 and 1000 times less poisonous. There are three different types of this toxin: A, B and C, of which toxins A and B are more toxic than C. Pumiliotoxins interfere with muscle contraction by affecting calcium channels, causing partial paralysis, difficulty moving, hyperactivity, or death. The median lethal dose of pumiliotoxins A and B is 50 μg / mouse, 20 μg / mouse respectively, while the amount of pumiliotoxin is 200 μg / frog.
Allopumiliotoxins are a structural division in the pumiliotoxin-A class of alkaloids. The compounds of the pumiliotoxin-A class are primarily found in the skins of frogs, toads, and other amphibians and are used as a chemical defense mechanism to ward off predators, microorganisms, and ectoparasites. The compounds were originally discovered in neotropical dendrobatid frogs, but are also found in the mantellid frogs of Madagascar, myobatrachid frogs of Australia, and bufonid toad of South America. Frogs possessing this defense mechanism have aposematic coloring.

Epiboxidine is a chemical compound which acts as a partial agonist at neural nicotinic acetylcholine receptors, binding to both the α3β4 and the α4β2 subtypes. It was developed as a less toxic analogue of the potent frog-derived alkaloid epibatidine, which is around 200 times stronger than morphine as an analgesic but produces extremely dangerous toxic nicotinic side effects.

Histrionicotoxins are a group of related toxins found in the skin of poison frogs from the family Dendrobatidae, notably Oophaga histrionica, which are native to Colombia. It is likely that, as with other poison frog alkaloids, histrionicotoxins are not manufactured by the amphibians, but absorbed from insects in their diet and stored in glands in their skin. They are notably less toxic than other alkaloids found in poison frogs, yet their distinct structure acts as a neurotoxin by non-competitive inhibition of nicotinic acetylcholine receptors.

Adelphobates is a small genus of poison dart frogs. They are found in the central and lower Amazon basin of Peru and Brazil, possibly Bolivia. It was originally erected as a sister group to the Dendrobates and Oophaga genera. The validity of the genus is still being discussed, with the alternative being "Dendrobates galactonotus group" within Dendrobates. One species originally placed in this genus as Adelphobates captivus has since been moved to the genus Excidobates erected in 2008.
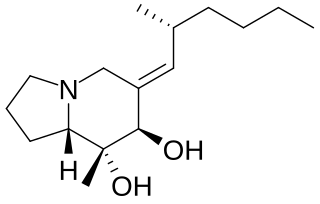
Gephyrotoxin is a naturally occurring product that stems from the Colombian tropical frog Dendrobates histrionicus. It is a member of the class of compounds known as histrionicotoxins. This alkaloid skin secretion was first isolated from the tropical frog in 1977 by Daly and his fellow workers.

Synthesis of morphine-like alkaloids in chemistry describes the total synthesis of the natural morphinan class of alkaloids that includes codeine, morphine, oripavine, and thebaine and the closely related semisynthetic analogs methorphan, buprenorphine, hydromorphone, hydrocodone, isocodeine, naltrexone, nalbuphine, oxymorphone, oxycodone, and naloxone.

Gelsemine (C20H22N2O2) is an indole alkaloid isolated from flowering plants of the genus Gelsemium, a plant native to the subtropical and tropical Americas, and southeast Asia, and is a highly toxic compound that acts as a paralytic, exposure to which can result in death. It has generally potent activity as an agonist of the mammalian glycine receptor, the activation of which leads to an inhibitory postsynaptic potential in neurons following chloride ion influx, and systemically, to muscle relaxation of varying intensity and deleterious effect. Despite its danger and toxicity, recent pharmacological research has suggested that the biological activities of this compound may offer opportunities for developing treatments related to xenobiotic or diet-induced oxidative stress, and of anxiety and other conditions, with ongoing research including attempts to identify safer derivatives and analogs to make use of gelsemine's beneficial effects.

















