The Scoville scale is a measurement of pungency of chili peppers and other substances, recorded in Scoville heat units (SHU). It is based on the concentration of capsaicinoids, among which capsaicin is the predominant component.
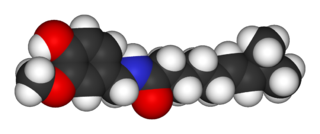
Capsaicin (8-methyl-N-vanillyl-6-nonenamide) is an active component of chili peppers, which are plants belonging to the genus Capsicum. It is a potent irritant for mammals, including humans, and produces a sensation of burning in any tissue with which it comes into contact. Capsaicin and several related amides (capsaicinoids) are produced as secondary metabolites by chili peppers, likely as deterrents against certain mammals and fungi. Pure capsaicin is a hydrophobic, colorless, highly pungent crystalline solid.
Euphorbium, an acrid dull-yellow or brown resin, consisting of the concreted milky juice of several species of Euphorbia, cactus-like perennial plants indigenous to Morocco. It dissolves in alcohol, ether and turpentine; in water it is only slightly soluble. It consists of two or more resins and a substance euphorbone, C20H36O or C15H24O. Pliny the Elder states that the name of the drug was given to it in honor of Euphorbus, the physician of Juba II, king of Mauretania. In former times euphorbium was valued in medicine for its drastic, purgative and emetic properties.

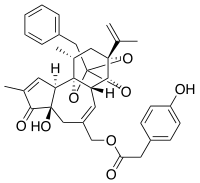
Resiniferatoxin (RTX) is a naturally occurring chemical found in resin spurge, a cactus-like plant commonly found in Morocco, and in Euphorbia poissonii found in northern Nigeria. It is a potent functional analog of capsaicin, the active ingredient in chili peppers.

Euphorbia resinifera, the resin spurge, is a species of spurge native to Morocco, where it occurs on the slopes of the Atlas Mountains. The dried latex of the plant was used in ancient medicine. It contains resiniferatoxin, an extremely potent capsaicin analog tested as an analgesic since 1997.

The transient receptor potential cation channel subfamily V member 1 (TRPV1), also known as the capsaicin receptor and the vanilloid receptor 1, is a protein that, in humans, is encoded by the TRPV1 gene. It was the first isolated member of the transient receptor potential vanilloid receptor proteins that in turn are a sub-family of the transient receptor potential protein group. This protein is a member of the TRPV group of transient receptor potential family of ion channels. Fatty acid metabolites with affinity for this receptor are produced by cyanobacteria, which diverged from eukaryotes at least 2000 million years ago (MYA). The function of TRPV1 is detection and regulation of body temperature. In addition, TRPV1 provides a sensation of scalding heat and pain (nociception). In primary afferent sensory neurons, it cooperates with TRPA1 to mediate the detection of noxious environmental stimuli.

Capsazepine is a synthetic antagonist of capsaicin. It is used as a biochemical tool in the study of TRPV ion channels.

Group C nerve fibers are one of three classes of nerve fiber in the central nervous system (CNS) and peripheral nervous system (PNS). The C group fibers are unmyelinated and have a small diameter and low conduction velocity, whereas Groups A and B are myelinated. Group C fibers include postganglionic fibers in the autonomic nervous system (ANS), and nerve fibers at the dorsal roots. These fibers carry sensory information.

Euphorbia poissonii, also known as Euphorbia poissoni and, incorrectly, as Euphorbia poisoni, is a highly irritant and toxic succulent member of the large and varied spurge family of plants. It is native to northern Nigeria, where local farmers extract its latex for use as a pesticide. Its powerfully irritant and pain-producing nature mandates use as a fencing plant. It is known to the Berom people of the Jos area as pyùlúp who transplant it to their compounds where it is regarded as protection against witchcraft.

Iodoresiniferatoxin (I-RTX) is a strong competitive antagonist of the Transient Receptor Potential Vanilloid 1 (TRPV1) receptor. I-RTX is derived from resiniferatoxin (RTX).
Relief from chronic pain remains a recognized unmet medical need. Consequently, the search for new analgesic agents is being intensively studied by the pharmaceutical industry. The TRPV1 receptor is a ligand gated ion channel that has been implicated in mediation of many types of pain and therefore studied most extensively. The first competitive antagonist, capsazepine, was first described in 1990; since then, several TRPV1 antagonists have entered clinical trials as analgesic agents. Should these new chemical entities relieve symptoms of chronic pain, then this class of compounds may offer one of the first novel mechanisms for the treatment of pain in many years.
Capsinoids are non-alkaloid substances naturally present in chili peppers. Although they are structurally similar to capsaicin, the substance that causes pungency in hot peppers, they largely lack that characteristic. Capsinoids have an estimated "hot taste threshold" which is about 1/1000 that of capsaicin. Capsinoids were not reported in the scientific literature until 1989, when biologists first isolated them in a unique variety of chili peppers, CH-19 Sweet, which does not contain capsaicin. Capsinoids include capsiate, dihydrocapsiate, and nordihydrocapsiate.
Zucapsaicin (Civanex) is a medication used to treat osteoarthritis of the knee and other neuropathic pain. Zucapsaicin is a member of phenols and a member of methoxybenzenes. It is a modulator of transient receptor potential cation channel subfamily V member 1 (TRPV-1), also known as the vanilloid or capsaicin receptor 1 that reduces pain, and improves articular functions. It is the cis-isomer of capsaicin. Civamide, manufactured by Winston Pharmaceuticals, is produced in formulations for oral, nasal, and topical use.

LASSBio-881 is a drug which acts as both a non-selective partial agonist of the CB1 and CB2 cannabinoid receptors, and also as an antagonist of the TRPV1 receptor, as well as having antioxidant effects. It has potent anti-inflammatory and anti-hyperalgesic effects in animal studies.

Vanillotoxins are neurotoxins found in the venom of the tarantula Psalmopoeus cambridgei. They act as agonists for the transient receptor potential cation channel subfamily V member 1 (TRPV1), activating the pain sensory system. VaTx1 and 2 also act as antagonists for the Kv2-type voltage-gated potassium channel (Kv2), inducing paralytic behavior in small animals.

The vanilloids are compounds which possess a vanillyl group. They include vanillyl alcohol, vanillin, vanillic acid, acetovanillon, vanillylmandelic acid, homovanillic acid, capsaicin, etc. Isomers are the isovanilloids.
RhTx is a small peptide toxin from Scolopendra subspinipes mutilans, also called the Chinese red-headed centipede. RhTx binds to the outer pore region of the temperature regulated TRPV1 ion channel, preferably in activated state, causing a downwards shift in the activation threshold temperature, which leads to the immediate onset of heat pain.

Phenylacetylrinvanil (IDN-5890) is a synthetic analogue of capsaicin which acts as a potent and selective agonist for the TRPV1 receptor, with slightly lower potency than resiniferatoxin, though still around 300 times the potency of capsaicin. It is an amide of vanillylamine and ricinoleic acid, with the hydroxyl group on ricinoleic acid esterified with phenylacetic acid. It is used to study the function of the TRPV1 receptor and its downstream actions, and has also shown anti-cancer effects in vitro.

SB-705498 is a drug which acts as a potent and selective blocker of the TRPV1 ion channel. It has been evaluated in clinical trials for the treatment of rhinitis and chronic cough.