
Heteropodatoxins are peptide toxins from the venom of the giant crab spider Heteropoda venatoria, which block Kv4.2 voltage-gated potassium channels.
Philanthotoxins are components of the venom of the Egyptian solitary wasp Philanthus triangulum, commonly known as the European beewolf. Philanthotoxins are polyamine toxins, a group of toxins isolated from the venom of wasps and spiders which immediately but reversibly paralyze their prey. δ-philanthotoxin, also known as PhTX-433, is the most active philanthotoxin that can be refined from the venom. PhTX-433 functions by non-selectively blocking excitatory neurotransmitter ion channels, including nicotinic acetylcholine receptors (nAChRs) and ionotropic glutamate receptors (iGluRs). Synthetic analogues, including PhTX-343 and PhTX-12, have been developed to improve selectivity. While the IC50 values of philanthotoxins varies between analogues and receptor subunit composition, the IC50 value of PhTX-433 at the iGluR AMPA receptor naturally expressed in locust leg muscle is 18 μM and the IC50 value at rat nAChRs is 1 μM.
BmTx3 is a neurotoxin, which is a component of the venom of the scorpion Buthus Martensi Karsch. It blocks A-type potassium channels in the central nervous system and hERG-channels in the heart.

Psalmotoxin (PcTx1) is a spider toxin from the venom of the Trinidad tarantula Psalmopoeus cambridgei. It selectively blocks Acid Sensing Ion Channel 1-a (ASIC1a), which is a proton-gated sodium channel.
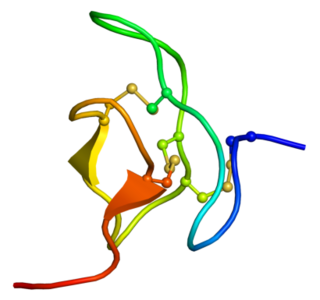
Phrixotoxins are peptide toxins derived from the venom of the Chilean copper tarantula Phrixotrichus auratus, also named Paraphysa scrofa. Phrixotoxin-1 and -2 block A-type voltage-gated potassium channels; phrixotoxin-3 blocks voltage-gated sodium channels. Similar toxins are found in other species, for instance the Chilean rose tarantula.
Jingzhaotoxin proteins are part of a venom secreted by Chilobrachys jingzhao, the Chinese tarantula. and act as neurotoxins. There are several subtypes of jingzhaotoxin, which differ in terms of channel selectivity and modification characteristics. All subspecies act as gating modifiers of sodium channels and/or, to a lesser extent, potassium channels.

Heteroscodratoxin-1 is a neurotoxin produced by the venom glands of Heteroscodra maculata that shifts the activation threshold of voltage-gated potassium channels and the inactivation of Nav1.1 sodium channels to more positive potentials.

delta-Palutoxins (δ-palutoxins) consist of a homologous group of four insect-specific toxins from the venom of the spider Pireneitega luctuosa. They show a high toxicity against Spodoptera litura larvae by inhibiting sodium channels, leading to strong paralytic activity and eventually to the death of the insect.

Guangxitoxin, also known as GxTX, is a peptide toxin found in the venom of the tarantula Plesiophrictus guangxiensis. It primarily inhibits outward voltage-gated Kv2.1 potassium channel currents, which are prominently expressed in pancreatic β-cells, thus increasing insulin secretion.
Hanatoxin is a toxin found in the venom of the Grammostola spatulata tarantula. The toxin is mostly known for inhibiting the activation of voltage-gated potassium channels, most specifically Kv4.2 and Kv2.1, by raising its activation threshold.
Theraphosa leblondi toxin (TLTx) is a toxin occurring in three different forms (subtypes) that are purified and sequenced from the venom of the giant tarantula Theraphosa blondi. This toxin selectively inhibits Kv4.2 voltage-gated potassium channels by acting as a gating modifier.
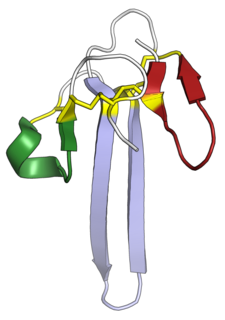
Mambalgins are peptides found in the venom of the black mamba, an elapid snake. Mambalgins are members of the three-finger toxin (3FTx) protein family and have the characteristic three-finger protein fold. First reported by French researchers in 2012, mambalgins are unusual members of the 3FTx family in that they have the in vivo effect of causing analgesia without apparent toxicity. Their mechanism of action is potent inhibition of acid-sensing ion channels.
Butantoxin (BuTX) is a compound of the venom of three Brazilian and an Argentinean scorpion species of the genus Tityus. Butantoxin reversibly blocks the voltage-gated K+ channels Shaker B and Kv1.2, and the Ca2+-activated K+ channelsKCa 1.1 and KCa 3.1.
Tamulotoxin is a venomous neurotoxin from the Indian Red Scorpion.
HgeTx1 (systematic name: α-KTx 6.14) is a toxin produced by the Mexican scorpion Hoffmanihadrurus gertschi that is a reversible blocker of the Shaker B K+-channel, a type of voltage-gated potassium channels.
RhTx is a small peptide toxin from Scolopendra subspinipes mutilans, also called the Chinese red-headed centipede. RhTx binds to the outer pore region of the temperature regulated TRPV1 ion channel, preferably in activated state, causing a downwards shift in the activation threshold temperature, which leads to the immediate onset of heat pain.
GTx1-15 is a toxin from the Chilean tarantula venom that acts as both a voltage-gated calcium channel blocker and a voltage-gated sodium channel blocker.
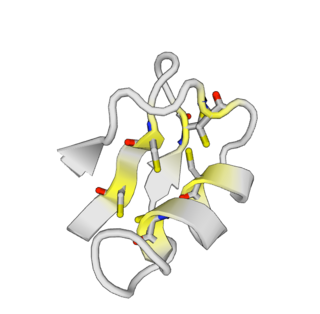
ImKTx88 is a selective inhibitor of the Kv1 ion channel family that can be isolated from the venom of the Isometrus maculatus. This peptide belongs to the α-KTx subfamily and is classified as a pore-blocking toxin.
LmαTX5 is an α-scorpion toxin which inhibits the fast inactivation of voltage-gated sodium channels. It has been identified through transcriptome analysis of the venom gland of Lychas mucronatus, also known as the Chinese swimming scorpion – a scorpion species which is widely distributed in Southeast Asia.
Protoxin-1, or Beta/omega-theraphotoxin-Tp1a, is a 35-amino-acid peptide neurotoxin extracted from the venom of the tarantula Thrixopelma pruriens. Protoxin-1 belongs to the inhibitory cystine knot (ICK) family of peptide toxins, which have been known to potently inhibit voltage-gated ion channels. Protoxin-1 selectively blocks low voltage threshold “T-type” calcium channels., voltage gated sodium channels and the nociceptor cation channel TRPA1. Due to its unique ability to bind to TRPA1, Protoxin-1 has been implicated as a valuable pharmacological reagent with potential applications in clinical contexts with regards to pain and inflammation








