| PA clan of proteases | |
|---|---|
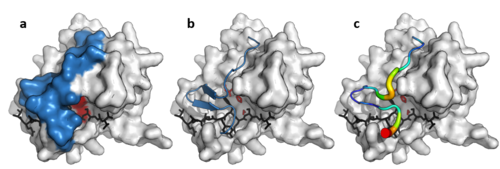
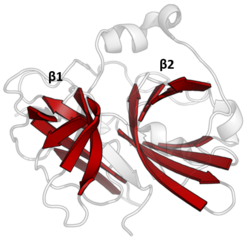 The double β-barrels that characterise the PA clan are highlighted in red. (TEV protease, PDB: 1lvm ) | |
| Identifiers | |
| Symbol | N/A |
| Pfam clan | CL0124 |
| ECOD | 1.1.5 |
| InterPro | IPR009003 |
| SCOP2 | 50494 / SCOPe / SUPFAM |
| Membranome | 319 |
The PA clan (Proteases of mixed nucleophile, superfamily A) is the largest group of proteases with common ancestry as identified by structural homology. Members have a chymotrypsin-like fold and similar proteolysis mechanisms but can have identity of <10%. The clan contains both cysteine and serine proteases (different nucleophiles). [1] [2] PA clan proteases can be found in plants, [3] animals, [3] fungi, [3] eubacteria, [4] archaea [5] [6] and viruses. [2]
Contents
- History
- Etymology
- Structure
- Double β-barrel
- Viral protease loop
- Evolution and function
- Catalytic activity
- Biological role and substrate specificity
- Families
- See also
- References
- External links
The common use of the catalytic triad for hydrolysis by multiple clans of proteases, including the PA clan, represents an example of convergent evolution. [7] The differences in the catalytic triad within the PA clan is also an example of divergent evolution of active sites in enzymes. [2]