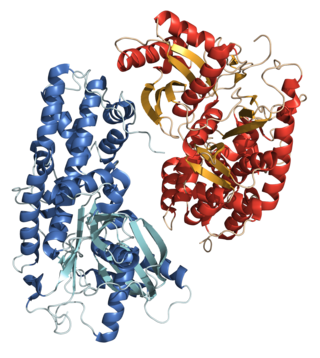
A hexokinase is an enzyme that phosphorylates hexoses, forming hexose phosphate. In most organisms, glucose is the most important substrate for hexokinases, and glucose-6-phosphate is the most important product. Hexokinase possesses the ability to transfer an inorganic phosphate group from ATP to a substrate.

In biochemistry, a phosphatase is an enzyme that uses water to cleave a phosphoric acid monoester into a phosphate ion and an alcohol. Because a phosphatase enzyme catalyzes the hydrolysis of its substrate, it is a subcategory of hydrolases. Phosphatase enzymes are essential to many biological functions, because phosphorylation and dephosphorylation serve diverse roles in cellular regulation and signaling. Whereas phosphatases remove phosphate groups from molecules, kinases catalyze the transfer of phosphate groups to molecules from ATP. Together, kinases and phosphatases direct a form of post-translational modification that is essential to the cell's regulatory network.
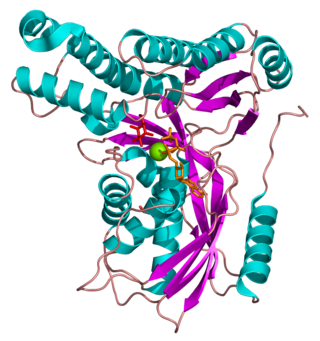
Galactokinase is an enzyme (phosphotransferase) that facilitates the phosphorylation of α-D-galactose to galactose 1-phosphate at the expense of one molecule of ATP. Galactokinase catalyzes the second step of the Leloir pathway, a metabolic pathway found in most organisms for the catabolism of α-D-galactose to glucose 1-phosphate. First isolated from mammalian liver, galactokinase has been studied extensively in yeast, archaea, plants, and humans.
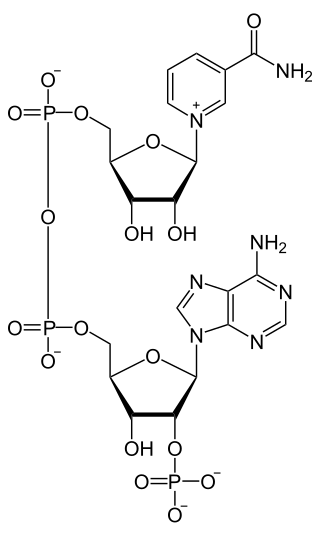
Nicotinamide adenine dinucleotide phosphate, abbreviated NADP+ or, in older notation, TPN (triphosphopyridine nucleotide), is a cofactor used in anabolic reactions, such as the Calvin cycle and lipid and nucleic acid syntheses, which require NADPH as a reducing agent ('hydrogen source'). NADPH is the reduced form of NADP+, the oxidized form. NADP+ is used by all forms of cellular life.

The enzyme Trehalase is a glycoside hydrolase, produced by cells in the brush border of the small intestine, which catalyzes the conversion of trehalose to glucose. It is found in most animals.
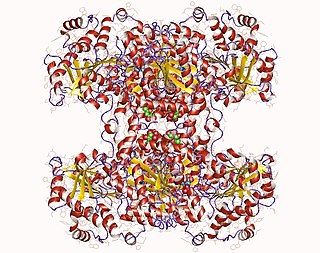
Glycogen synthase is a key enzyme in glycogenesis, the conversion of glucose into glycogen. It is a glycosyltransferase that catalyses the reaction of UDP-glucose and n to yield UDP and n+1.

The enzyme glucose 6-phosphatase (EC 3.1.3.9, G6Pase; systematic name D-glucose-6-phosphate phosphohydrolase) catalyzes the hydrolysis of glucose 6-phosphate, resulting in the creation of a phosphate group and free glucose:

2-Deoxy-d-glucose is a glucose molecule which has the 2-hydroxyl group replaced by hydrogen, so that it cannot undergo further glycolysis. As such; it acts to competitively inhibit the production of glucose-6-phosphate from glucose at the phosphoglucoisomerase level. 2-Deoxyglucose labeled with tritium or carbon-14 has been a popular ligand for laboratory research in animal models, where distribution is assessed by tissue-slicing followed by autoradiography, sometimes in tandem with either conventional or electron microscopy.
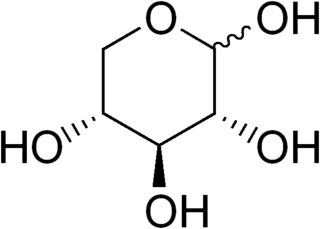
D-Xylose is a five-carbon aldose that can be catabolized or metabolized into useful products by a variety of organisms.
In enzymology, a phosphogluconate dehydrogenase (decarboxylating) (EC 1.1.1.44) is an enzyme that catalyzes the chemical reaction
CDP-4-dehydro-6-deoxyglucose reductase (EC 1.17.1.1) is an enzyme that catalyzes the chemical reaction
In enzymology, an inositol-3-phosphate synthase is an enzyme that catalyzes the chemical reaction
In enzymology, a phosphopentomutase is an enzyme that catalyzes the chemical reaction
The enzyme glycerol-1-phosphatase (EC 3.1.3.21) catalyzes the reaction

In enzymology, glucosamine-phosphate N-acetyltransferase (GNA) is an enzyme that catalyzes the transfer of an acetyl group from acetyl-CoA to the primary amine in glucosamide-6-phosphate, generating a free CoA and N-acetyl-D-glucosamine-6-phosphate.
In enzymology, a dTDP-4-amino-4,6-dideoxy-D-glucose transaminase is an enzyme that catalyzes the chemical reaction

Glucose-6-phosphatase, catalytic subunit is an enzyme that in humans is encoded by the G6PC gene.
3-Deoxy-D-arabinoheptulosonate 7-phosphate (DAHP) synthase is the first enzyme in a series of metabolic reactions known as the shikimate pathway, which is responsible for the biosynthesis of the amino acids phenylalanine, tyrosine, and tryptophan. Since it is the first enzyme in the shikimate pathway, it controls the amount of carbon entering the pathway. Enzyme inhibition is the primary method of regulating the amount of carbon entering the pathway. Forms of this enzyme differ between organisms, but can be considered DAHP synthase based upon the reaction that is catalyzed by this enzyme.
Diacylglycerol diphosphate phosphatase (EC 3.1.3.81, DGPP phosphatase, DGPP phosphohydrolase, DPP1, DPPL1, DPPL2, PAP2, pyrophosphate phosphatase) is an enzyme with systematic name 1,2-diacyl-sn-glycerol 3-phosphate phosphohydrolase. This enzyme catalyses the following chemical reaction
ADP-ribose 1′′-phosphate phosphatase (EC 3.1.3.84, POA1, Appr1p phosphatase, Poa1p) is an enzyme with systematic name ADP-D-ribose 1′′-phosphate phosphohydrolase. This enzyme catalyses the following chemical reaction














