
Type 2 diabetes (T2D), formerly known as adult-onset diabetes, is a form of diabetes that is characterized by high blood sugar, insulin resistance, and relative lack of insulin. Common symptoms include increased thirst, frequent urination, and unexplained weight loss. Symptoms may also include increased hunger, feeling tired, and sores that do not heal. Often symptoms come on slowly. Long-term complications from high blood sugar include heart disease, strokes, diabetic retinopathy which can result in blindness, kidney failure, and poor blood flow in the limbs which may lead to amputations. The sudden onset of hyperosmolar hyperglycemic state may occur; however, ketoacidosis is uncommon.
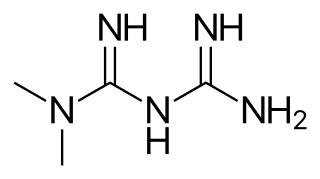
Metformin, marketed under the trade name Glucophage among others, is the first-line medication for the treatment of type 2 diabetes, particularly in people who are overweight. It is also used in the treatment of polycystic ovary syndrome. It is not associated with weight gain. It is taken by mouth.

Rosiglitazone is an antidiabetic drug in the thiazolidinedione class. It works as an insulin sensitizer, by binding to the PPAR in fat cells and making the cells more responsive to insulin. It is marketed by the pharmaceutical company GlaxoSmithKline (GSK) as a stand-alone drug or for use in combination with metformin or with glimepiride. First released in 1999, annual sales peaked at approximately $2.5-billion in 2006; however, following a meta-analysis in 2007 that linked the drug's use to an increased risk of heart attack, sales plummeted to just $9.5-million in 2012. The drug's patent expired in 2012.

Pioglitazone, sold under the brand name Actos among others, is an anti-diabetic medication used to treat type 2 diabetes. It may be used with metformin, a sulfonylurea, or insulin. Use is recommended together with exercise and diet. It is not recommended in type 1 diabetes. It is taken by mouth.
Sulfonylureas are a class of organic compounds used in medicine and agriculture, for example as antidiabetic drugs widely used in the management of diabetes mellitus type 2. They act by increasing insulin release from the beta cells in the pancreas. A number of sulfonylureas are also used as herbicides, because they can interfere with plant biosynthesis of certain amino acids. Sulfonylureas are also used experimentally to inhibit interleukin 1 beta release from the NALP3 inflammasome.

Diabetic nephropathy, also known as diabetic kidney disease, is the chronic loss of kidney function occurring in those with diabetes mellitus. Diabetic nephropathy is one of the leading causes of chronic kidney disease (CKD) and end-stage renal disease (ESRD) globally. Protein loss in the urine due to damage to the glomeruli may become massive, and cause a low serum albumin with resulting generalized body swelling (edema) and result in the nephrotic syndrome. Likewise, the estimated glomerular filtration rate (eGFR) may progressively fall from a normal of over 90 ml/min/1.73m2 to less than 15, at which point the patient is said to have end-stage renal disease. It usually is slowly progressive over years.

Exenatide, sold under the brand name Byetta and Bydureon among others, is a medication used to treat diabetes mellitus type 2. It is used together with diet, exercise, and potentially other antidiabetic medication. It is a treatment option after metformin and sulfonylureas. It is given by injection under the skin within an hour before the first and last meal of the day. A once-weekly injection version is also available.

Sitagliptin, sold under the brand name Januvia among others, is an anti-diabetic medication used to treat type 2 diabetes. In the United Kingdom it is listed as less preferred than metformin or a sulfonylurea. It is taken by mouth. It is also available in the fixed-dose combination medication sitagliptin/metformin.

Saxagliptin, sold under the brand name Onglyza, is an oral hypoglycemic of the dipeptidyl peptidase-4 (DPP-4) inhibitor class. Early development was solely by Bristol-Myers Squibb; in 2007 AstraZeneca joined with Bristol-Myers Squibb to co-develop the final compound and collaborate on the marketing of the drug.
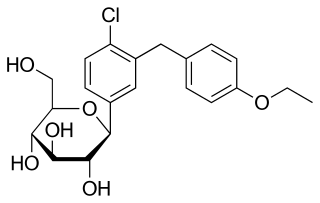
Dapagliflozin, sold under the brand name Farxiga among others, is a medication used to treat type 2 diabetes and, with certain restrictions, type 1 diabetes. It is also used to treat adults with heart failure with reduced ejection fraction to reduce the risk of cardiovascular death and hospitalization for heart failure.

Liraglutide, sold under the brand name Victoza among others, is a medication used to treat diabetes mellitus type 2 and obesity. In diabetes it is a less preferred agent compared to metformin. Its effects on long-term health outcomes like heart disease and life expectancy are unclear. It is given by injection under the skin.
Albiglutide is a glucagon-like peptide-1 agonist drug marketed by GlaxoSmithKline (GSK) for treatment of type 2 diabetes. As of 2017 it is unclear if it affects a person's risk of death. GSK has announced that it intends to withdraw the drug from the worldwide market by July 2018 for economic reasons.
Glucagon-like peptide-1 receptor agonists, also known as GLP-1 receptor agonists or incretin mimetics, are agonists of the GLP-1 receptor. This class of medications is used for the treatment of type 2 diabetes. One of their advantages over older insulin secretagogues, such as sulfonylureas or meglitinides, is that they have a lower risk of causing hypoglycemia. GLP-1 has a short duration of action, so to overcome this limitation several modifications either in the drug or the formulations are being developed.

Canagliflozin, sold under the brand name Invokana among others, is a medication used to treat type 2 diabetes. It is a third-line medication to metformin. It is used together with exercise and diet. It is not recommended in type 1 diabetes. It is taken by mouth.
Andrej Janež is a Slovenian diabetologist and diabetes researcher. Janež is the Head of Department of Endocrinology, Diabetes and Metabolic Disease at University Medical Centre Ljubljana, Assistant Professor for Internal Medicine at the Medical University Ljubljana, Chairman of the Advances in Diabetes and Insulin Therapy conference, member of the advisory board for peroral antidiabetic therapy in Servier Pharma, member for Slovenia in the Diabetes Education Study Group at European Association for the Study of Diabetes, and member of the European advisory board for continuous glucose monitoring system in development for Lifescan.
Empagliflozin, sold under the brand name Jardiance among others, is a medication used together with diet and exercise to treat type 2 diabetes. It can be prescribed instead of metformin and has benefits over sulfonylureas. It may be used together with other medications such as metformin or insulin. It is not recommended for type 1 diabetes. It is taken by mouth. In view of recent trial evidence, it is expected to soon receive licence to be used for patients with heart failure, irrespective of diabetic status.
Dulaglutide, sold under the brand name Trulicity among others, is a medication used for the treatment of type 2 diabetes in combination with diet and exercise. It is also approved in the United States for the reduction of major adverse cardiovascular events in adults with type 2 diabetes who have established cardiovascular disease or multiple cardiovascular risk factors. It can be used once weekly.
Gliflozins are a class of drugs in the treatment of type 2 diabetes (T2D). They act by inhibiting sodium/glucose cotransporter 2 (SGLT-2), and are therefore also called SGLT-2 inhibitors. The efficacy of the drug is dependent on renal excretion and prevents glucose from going into blood circulation by promoting glucosuria. The mechanism of action is insulin independent.
SGLT2 inhibitors, also called gliflozins, are a class of medications that alter essential physiology of the nephron; unlike SGLT1 inhibitors that modulate sodium/glucose channels in the intestinal mucosa. All of these advances are within the influence of the #SLC5A gene family. The foremost metabolic effect appears to show that this pharmaceutical class inhibits reabsorption of glucose in the kidney and therefore lower blood sugar. They act by inhibiting sodium-glucose transport protein 2 (SGLT2). SGLT2 inhibitors are used in the treatment of type II diabetes mellitus (T2DM). Apart from blood sugar control, gliflozins have been shown to provide significant cardiovascular benefit in T2DM patients. Several medications of this class have been approved or are currently under development. In studies on canagliflozin, a member of this class, the medication was found to enhance blood sugar control as well as reduce body weight and systolic and diastolic blood pressure.

Ipragliflozin is a pharmaceutical drug for treatment of type 2 diabetes. Ipragliflozin, jointly developed by Astellas Pharma and Kotobuki Pharmaceutical, was approved in Japan on January 17, 2014, and in Russia on May 22, 2019.












