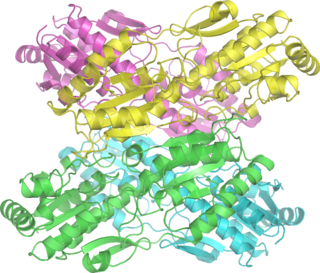
Phosphofructokinase-1 (PFK-1) is one of the most important regulatory enzymes of glycolysis. It is an allosteric enzyme made of 4 subunits and controlled by many activators and inhibitors. PFK-1 catalyzes the important "committed" step of glycolysis, the conversion of fructose 6-phosphate and ATP to fructose 1,6-bisphosphate and ADP. Glycolysis is the foundation for respiration, both anaerobic and aerobic. Because phosphofructokinase (PFK) catalyzes the ATP-dependent phosphorylation to convert fructose-6-phosphate into fructose 1,6-bisphosphate and ADP, it is one of the key regulatory steps of glycolysis. PFK is able to regulate glycolysis through allosteric inhibition, and in this way, the cell can increase or decrease the rate of glycolysis in response to the cell's energy requirements. For example, a high ratio of ATP to ADP will inhibit PFK and glycolysis. The key difference between the regulation of PFK in eukaryotes and prokaryotes is that in eukaryotes PFK is activated by fructose 2,6-bisphosphate. The purpose of fructose 2,6-bisphosphate is to supersede ATP inhibition, thus allowing eukaryotes to have greater sensitivity to regulation by hormones like glucagon and insulin.
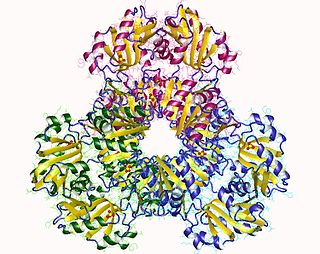
Ribose-phosphate diphosphokinase is an enzyme that converts ribose 5-phosphate into phosphoribosyl pyrophosphate (PRPP). It is classified under EC 2.7.6.1.
Arsenate reductase (glutaredoxin) (EC 1.20.4.1) is an enzyme that catalyzes the chemical reaction
The enzyme sabinene-hydrate synthase (EC 4.2.3.11) catalyzes the chemical reaction
In enzymology, a Cd2+-exporting ATPase (EC 3.6.3.3) is an enzyme that catalyzes the chemical reaction
In enzymology, a Cl-transporting ATPase (EC 3.6.3.11) is an enzyme that catalyzes the chemical reaction
In enzymology, a FAD diphosphatase (EC 3.6.1.18) is an enzyme that catalyzes the chemical reaction
In enzymology, a nucleoside-triphosphatase(NTPase) (EC 3.6.1.15) is an enzyme that catalyzes the chemical reaction
In enzymology, a nucleotide diphosphatase (EC 3.6.1.9) is an enzyme that catalyzes the chemical reaction
In enzymology, a Zn2+-exporting ATPase (EC 3.6.3.5) is an enzyme that catalyzes the chemical reaction
The enzyme dodecanoyl-[acyl-carrier-protein] hydrolase (EC 3.1.2.21) catalyzes the reaction
The enzyme [hydroxymethylglutaryl-CoA reductase (NADPH)]-phosphatase (EC 3.1.3.47) catalyzes the reaction
The enzyme pyridoxal phosphatase (EC 3.1.3.74) catalyzes the reaction
The enzyme [pyruvate dehydrogenase (acetyl-transferring)]-phosphatase (EC 3.1.3.43) catalyzes the reaction

In enzymology, a guanylate kinase is an enzyme that catalyzes the chemical reaction
In enzymology, a [isocitrate dehydrogenase (NADP+)] kinase (EC 2.7.11.5) is an enzyme that catalyzes the chemical reaction:
In enzymology, a ribose-5-phosphate adenylyltransferase is an enzyme that catalyzes the chemical reaction

In molecular biology, the ars operon is an operon found in several bacterial taxon. It is required for the detoxification of arsenate, arsenite, and antimonite. This system transports arsenite and antimonite out of the cell. The pump is composed of two polypeptides, the products of the arsA and arsB genes. This two-subunit enzyme produces resistance to arsenite and antimonite. Arsenate, however, must first be reduced to arsenite before it is extruded. A third gene, arsC, expands the substrate specificity to allow for arsenate pumping and resistance. ArsC is an approximately 150-residue arsenate reductase that uses reduced glutathione (GSH) to convert arsenate to arsenite with a redox active cysteine residue in the active site. ArsC forms an active quaternary complex with GSH, arsenate, and glutaredoxin 1 (Grx1). The three ligands must be present simultaneously for reduction to occur.
Arsenite resistance (Ars) efflux pumps of bacteria may consist of two proteins, ArsB and ArsA, or of one protein. ArsA proteins have two ATP binding domains and probably arose by a tandem gene duplication event. ArsB proteins all possess twelve transmembrane spanners and may also have arisen by a tandem gene duplication event. Structurally, the Ars pumps resemble ABC-type efflux pumps, but there is no significant sequence similarity between the Ars and ABC pumps. When only ArsB is present, the system operates by a pmf-dependent mechanism, and consequently belongs in TC subclass 2.A. When ArsA is also present, ATP hydrolysis drives efflux, and consequently the system belongs in TC subclass 3.A. ArsB therefore appears twice in the TC system but ArsA appears only once. These pumps actively expel both arsenite and antimonite.
Daniel Edward Atkinson is an American biochemist who worked at UCLA for 40 years from 1952 until his retirement in 1992, though he continued his scientific work as Emeritus Professor. He is best known for the concept of energy charge.




