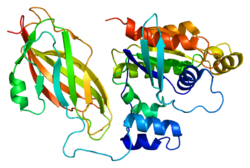
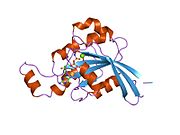
Glucose transporter type 4 (GLUT4), also known as solute carrier family 2, facilitated glucose transporter member 4, is a protein encoded, in humans, by the SLC2A4 gene. GLUT4 is the insulin-regulated glucose transporter found primarily in adipose tissues and striated muscle. The first evidence for this distinct glucose transport protein was provided by David James in 1988. The gene that encodes GLUT4 was cloned and mapped in 1989.

Guanine nucleotide exchange factors (GEFs) are proteins or protein domains that activate monomeric GTPases by stimulating the release of guanosine diphosphate (GDP) to allow binding of guanosine triphosphate (GTP). A variety of unrelated structural domains have been shown to exhibit guanine nucleotide exchange activity. Some GEFs can activate multiple GTPases while others are specific to a single GTPase.
The Rho family of GTPases is a family of small signaling G proteins, and is a subfamily of the Ras superfamily. The members of the Rho GTPase family have been shown to regulate many aspects of intracellular actin dynamics, and are found in all eukaryotic kingdoms, including yeasts and some plants. Three members of the family have been studied in detail: Cdc42, Rac1, and RhoA. All G proteins are "molecular switches", and Rho proteins play a role in organelle development, cytoskeletal dynamics, cell movement, and other common cellular functions.

Cell division control protein 42 homolog is a protein that in humans is encoded by the CDC42 gene. Cdc42 is involved in regulation of the cell cycle. It was originally identified in S. cerevisiae (yeast) as a mediator of cell division, and is now known to influence a variety of signaling events and cellular processes in a variety of organisms from yeast to mammals.

Transforming protein RhoA, also known as Ras homolog family member A (RhoA), is a small GTPase protein in the Rho family of GTPases that in humans is encoded by the RHOA gene. While the effects of RhoA activity are not all well known, it is primarily associated with cytoskeleton regulation, mostly actin stress fibers formation and actomyosin contractility. It acts upon several effectors. Among them, ROCK1 and DIAPH1 are the best described. RhoA, and the other Rho GTPases, are part of a larger family of related proteins known as the Ras superfamily, a family of proteins involved in the regulation and timing of cell division. RhoA is one of the oldest Rho GTPases, with homologues present in the genomes since 1.5 billion years. As a consequence, RhoA is somehow involved in many cellular processes which emerged throughout evolution. RhoA specifically is regarded as a prominent regulatory factor in other functions such as the regulation of cytoskeletal dynamics, transcription, cell cycle progression and cell transformation.

Serine/threonine-protein kinase PAK 1 is an enzyme that in humans is encoded by the PAK1 gene.

ROCK1 is a protein serine/threonine kinase also known as rho-associated, coiled-coil-containing protein kinase 1. Other common names are ROKβ and P160ROCK. ROCK1 is a major downstream effector of the small GTPase RhoA and is a regulator of the actomyosin cytoskeleton which promotes contractile force generation. ROCK1 plays a role in cancer and in particular cell motility, metastasis, and angiogenesis.

Rac GTPase-activating protein 1 is an enzyme that in humans is encoded by the RACGAP1 gene.

Rho guanine nucleotide exchange factor TIAM1 is a protein that in humans is encoded by the TIAM1 gene.

Ras GTPase-activating-like protein IQGAP1 (IQGAP1) also known as p195 is a ubiquitously expressed protein that in humans is encoded by the IQGAP1 gene. IQGAP1 is a scaffold protein involved in regulating various cellular processes ranging from organization of the actin cytoskeleton, transcription, and cellular adhesion to regulating the cell cycle.
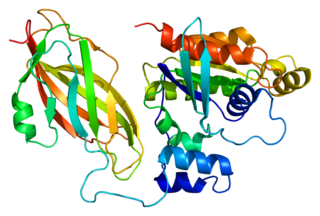
Rac2 is a small signaling G protein, and is a member of the Rac subfamily of the family Rho family of GTPases. It is encoded by the gene RAC2.

RhoC is a small signaling G protein, and is a member of the Rac subfamily of the family Rho family of GTPases. It is encoded by the gene RHOC.

Rho guanine nucleotide exchange factor 6 is a protein that, in humans, is encoded by the ARHGEF6 gene.

Dedicator of cytokinesis protein 1 (Dock1), also (DOCK180), is a large protein encoded in the human by the DOCK1 gene, involved in intracellular signalling networks. It is the mammalian ortholog of the C. elegans protein CED-5 and belongs to the DOCK family of guanine nucleotide exchange factors (GEFs).

Rho GDP-dissociation inhibitor 1 is a protein that in humans is encoded by the ARHGDIA gene.

Rho GDP-dissociation inhibitor 2 is a protein that in humans is encoded by the ARHGDIB gene. Aliases of this gene include RhoGDI2, GDID4, Rho GDI 2, and others.

RhoG is a small monomeric GTP-binding protein, and is an important component of many intracellular signalling pathways. It is a member of the Rac subfamily of the Rho family of small G proteins and is encoded by the gene RHOG.
Triple functional domain protein is a protein that in humans is encoded by the TRIO gene.

Rho GTPase-activating protein 32 is a protein that in humans is encoded by the RICS gene. RICS has two known isoforms, RICS that are expressed primarily at neurite growth cones, and at the post synaptic membranes, and PX-RICS which is more widely expressed in the endoplasmic reticulum, Golgi apparatus and endosomes. The only known domain of the RICS is the RhoGAP domain, whilst PX-RICS has an additional Phox homology and SH3 domain.

Dedicator of cytokinesis protein 4 (Dock4), is a large protein encoded in the human by the DOCK4 gene, involved in intracellular signalling networks. It is a member of the DOCK-B subfamily of the DOCK family of guanine nucleotide exchange factors (GEFs) which function as activators of small G-proteins. Dock4 activates the small G proteins Rac and Rap1.