Contents
- Biosynthesis and occurrence
- Uses
- Food
- Medicine
- Other uses
- Synthesis
- Historic and laboratory routes
- Reactions
- Safety
- See also
- References
- External links
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name (2E)-But-2-enedioic acid | |
Other names
| |
| Identifiers | |
3D model (JSmol) | |
| 605763 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.003.404 |
| EC Number |
|
| E number | E297 (preservatives) |
| 49855 | |
| KEGG | |
PubChem CID | |
| RTECS number |
|
| UNII | |
| UN number | 9126 |
CompTox Dashboard (EPA) | |
| |
| |
| Properties | |
| C4H4O4 | |
| Molar mass | 116.072 g·mol−1 |
| Appearance | White solid |
| Density | 1.635 g/cm3 |
| Melting point | 287 °C (549 °F; 560 K) (decomposes) [1] |
| 6.3 g/L at 25 °C [2] | |
| Acidity (pKa) | pka1 = 3.03, pka2 = 4.44 (15 °C, cis isomer) |
| −49.11·10−6 cm3/mol | |
| non zero | |
| Pharmacology | |
| D05AX01 ( WHO ) | |
| Hazards | |
| GHS labelling: | |
 | |
| Warning | |
| H319 | |
| P264, P280, P305+P351+P338, P313 | |
| NFPA 704 (fire diamond) | |
| 375 °C (707 °F; 648 K) | |
| Related compounds | |
Related carboxylic acids | |
Related compounds |
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
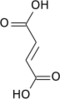
Fumaric acid or trans-butenedioic acid is an organic compound with the formula HO2CCH=CHCO2H. A white solid, fumaric acid occurs widely in nature. It has a fruit-like taste and has been used as a food additive. Its E number is E297. [3] The salts and esters are known as fumarates. Fumarate can also refer to the C
4H
2O2−
4 ion (in solution). Fumaric acid is the trans isomer of butenedioic acid, while maleic acid is the cis isomer.