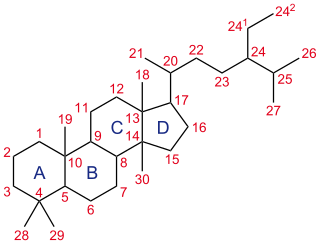
A steroid is a biologically active organic compound with four rings arranged in a specific molecular configuration. Steroids have two principal biological functions: as important components of cell membranes that alter membrane fluidity; and as signaling molecules. Hundreds of steroids are found in plants, animals and fungi. All steroids are manufactured in cells from the sterols lanosterol (opisthokonts) or cycloartenol (plants). Lanosterol and cycloartenol are derived from the cyclization of the triterpene squalene.

A steroid hydroxylase is a class of hydroxylase enzymes involved in the biosynthesis of steroids.
3β-Hydroxysteroid dehydrogenase/Δ5-4 isomerase (3β-HSD) is an enzyme that catalyzes the biosynthesis of the steroid progesterone from pregnenolone, 17α-hydroxyprogesterone from 17α-hydroxypregnenolone, and androstenedione from dehydroepiandrosterone (DHEA) in the adrenal gland. It is the only enzyme in the adrenal pathway of corticosteroid synthesis that is not a member of the cytochrome P450 family. It is also present in other steroid-producing tissues, including the ovary, testis and placenta. In humans, there are two 3β-HSD isozymes encoded by the HSD3B1 and HSD3B2 genes.

Steroid 21-hydroxylase is an enzyme that hydroxylates steroids at the C21 position and is involved in biosynthesis of aldosterone and cortisol. The enzyme converts progesterone and 17α-hydroxyprogesterone into 11-deoxycorticosterone and 11-deoxycortisol, respectively, within metabolic pathways that ultimately lead to aldosterone and cortisol. Deficiency in the enzyme may cause congenital adrenal hyperplasia.
In enzymology, a 3-oxo-5beta-steroid 4-dehydrogenase is an enzyme that catalyzes the chemical reaction
The enzyme 1,4-lactonase (EC 3.1.1.25) catalyzes the generic reaction
The enzyme 3-oxoadipate enol-lactonase (EC 3.1.1.24) catalyzes the reaction
The enzyme actinomycin lactonase (EC 3.1.1.39) catalyzes the reaction
The enzyme arylesterase (EC 3.1.1.2) catalyzes the reaction
The enzyme deoxylimonate A-ring-lactonase (EC 3.1.1.46) catalyzes the reaction
The enzyme gluconolactonase (EC 3.1.1.17) catalyzes the reaction
The enzyme L-rhamnono-1,4-lactonase (EC 3.1.1.65) catalyzes the reaction
The enzyme triacetate-lactonase (EC 3.1.1.38) catalyzes the reaction
The enzyme xylono-1,4-lactonase (EC 3.1.1.68) catalyzes the reaction

The alpha/beta hydrolase superfamily is a superfamily of hydrolytic enzymes of widely differing phylogenetic origin and catalytic function that share a common fold. The core of each enzyme is an alpha/beta-sheet, containing 8 beta strands connected by 6 alpha helices. The enzymes are believed to have diverged from a common ancestor, retaining little obvious sequence similarity, but preserving the arrangement of the catalytic residues. All have a catalytic triad, the elements of which are borne on loops, which are the best-conserved structural features of the fold.

Lactonase (EC 3.1.1.81, acyl-homoserine lactonase; systematic name N-acyl-L-homoserine-lactone lactonohydrolase) is a metalloenzyme, produced by certain species of bacteria, which targets and inactivates acylated homoserine lactones (AHLs). It catalyzes the reaction

Paraoxonases are a family of mammalian enzymes with aryldialkylphosphatase activity. There are three paraoxonase isozymes, which were originally discovered for their involvement in the hydrolysis of organophosphates.
Ketosteroid monooxygenase (EC 1.14.13.54, steroid-ketone monooxygenase, progesterone, NADPH2:oxygen oxidoreductase (20-hydroxylating, ester-producing), 17alpha-hydroxyprogesterone, NADPH2:oxygen oxidoreductase (20-hydroxylating, side-chain cleaving), androstenedione, NADPH2:oxygen oxidoreductase (17-hydroxylating, lactonizing)) is an enzyme with systematic name ketosteroid,NADPH:oxygen oxidoreductase (20-hydroxylating, ester-producing/20-hydroxylating, side-chain cleaving/17-hydroxylating, lactonizing). This enzyme catalyses the following chemical reaction
The enzyme 2-oxo-3-(5-oxofuran-2-ylidene)propanoate lactonase (EC 3.1.1.91, naaC (gene); systematic name 2-oxo-3-(5-oxofuran-2-ylidene)propanoate lactonohydrolase) catalyses the reaction

5α-Dihydronorethisterone is a major active metabolite of norethisterone (norethindrone). Norethisterone is a progestin with additional weak androgenic and estrogenic activity. 5α-DHNET is formed from norethisterone by 5α-reductase in the liver and other tissues.









