| 2-oxo-3-(5-oxofuran-2-ylidene)propanoate lactonase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||
| EC no. | 3.1.1.91 | ||||||||
| Databases | |||||||||
| IntEnz | IntEnz view | ||||||||
| BRENDA | BRENDA entry | ||||||||
| ExPASy | NiceZyme view | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| |||||||||
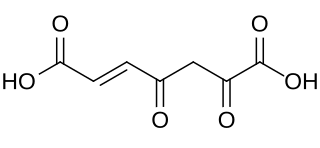
The enzyme 2-oxo-3-(5-oxofuran-2-ylidene)propanoate lactonase (EC 3.1.1.91, naaC (gene); systematic name 2-oxo-3-(5-oxofuran-2-ylidene)propanoate lactonohydrolase) [1] catalyses the reaction
- 2-oxo-3-(5-oxofuran-2-ylidene)propanoate + H2O maleylpyruvate
This enzyme is isolated from the soil bacterium Bradyrhizobium sp. JS329.