Immunotherapy or biological therapy is the treatment of disease by activating or suppressing the immune system. Immunotherapies designed to elicit or amplify an immune response are classified as activation immunotherapies, while immunotherapies that reduce or suppress are classified as suppression immunotherapies. Immunotherapy is under preliminary research for its potential to treat various forms of cancer.

Immunosuppressive drugs, also known as immunosuppressive agents, immunosuppressants and antirejection medications, are drugs that inhibit or prevent activity of the immune system.
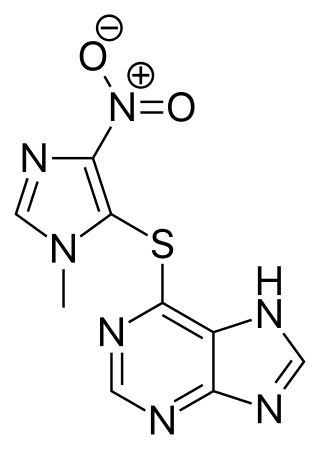
Azathioprine, sold under the brand name Imuran, among others, is an immunosuppressive medication. It is used for the treatment of rheumatoid arthritis, granulomatosis with polyangiitis, Crohn's disease, ulcerative colitis, and systemic lupus erythematosus; and in kidney transplants to prevent rejection. It is listed by the International Agency for Research on Cancer as a group 1 human carcinogen. It is taken by mouth or injected into a vein.
Daclizumab is a therapeutic humanized monoclonal antibody which was used for the treatment of adults with relapsing forms of multiple sclerosis (MS). Daclizumab works by binding to CD25, the alpha subunit of the IL-2 receptor of T-cells.

Elan Corporation plc was a major drugs firm based in Dublin, Ireland, which had major interests in the United States. It was listed on the New York Stock Exchange as ELN, the Irish Stock Exchange as ELN.I, and the London Stock Exchange as ELN.L. In 2013, the company merged with Perrigo to form Perrigo Company PLC.

Rituximab, sold under the brand name Rituxan among others, is a monoclonal antibody medication used to treat certain autoimmune diseases and types of cancer. It is used for non-Hodgkin lymphoma, chronic lymphocytic leukemia, rheumatoid arthritis, granulomatosis with polyangiitis, idiopathic thrombocytopenic purpura, pemphigus vulgaris, myasthenia gravis and Epstein–Barr virus-positive mucocutaneous ulcers. It is given by slow injection into a vein. Biosimilars of Rituxan include Blitzima, Riabni, Ritemvia, Rituenza, Rixathon, Ruxience, and Truxima.

Natalizumab, sold under the brand name Tysabri among others, is a medication used to treat multiple sclerosis and Crohn's disease. It is a humanized monoclonal antibody against the cell adhesion molecule α4-integrin. It is given by intravenous infusion every 28 days. The drug is believed to work by reducing the ability of inflammatory immune cells to attach to and pass through the cell layers lining the intestines and blood–brain barrier. Natalizumab has proven effective in treating the symptoms of both diseases, preventing relapse, vision loss, cognitive decline and significantly improving quality of life in people with multiple sclerosis, as well as increasing rates of remission and preventing relapse in multiple sclerosis.

Alemtuzumab, sold under the brand names Campath and Lemtrada among others, is a medication used to treat chronic lymphocytic leukemia (CLL) and multiple sclerosis. In CLL, it has been used as both a first line and second line treatment. In MS it is generally only recommended if other treatments have not worked. It is given by injection into a vein.
A TNF inhibitor is a pharmaceutical drug that suppresses the physiologic response to tumor necrosis factor (TNF), which is part of the inflammatory response. TNF is involved in autoimmune and immune-mediated disorders such as rheumatoid arthritis, ankylosing spondylitis, inflammatory bowel disease, psoriasis, hidradenitis suppurativa and refractory asthma, so TNF inhibitors may be used in their treatment. The important side effects of TNF inhibitors include lymphomas, infections, congestive heart failure, demyelinating disease, a lupus-like syndrome, induction of auto-antibodies, injection site reactions, and systemic side effects.
Management of Crohn's disease involves first treating the acute symptoms of the disease, then maintaining remission. Since Crohn's disease is an immune system condition, it cannot be cured by medication or surgery. Treatment initially involves the use of medications to eliminate infections and reduce inflammation. Surgery may be required for complications such as obstructions or abscesses, or if the disease does not respond to drugs within a reasonable time. However, surgery cannot cure Crohn's disease. It involves removing the diseased part of the intestine and rejoining the healthy ends, but the disease tends to recur after surgery.

Biological therapy, the use of medications called biopharmaceuticals or biologics that are tailored to specifically target an immune or genetic mediator of disease, plays a major role in the treatment of inflammatory bowel disease. Even for diseases of unknown cause, molecules that are involved in the disease process have been identified, and can be targeted for biological therapy. Many of these molecules, which are mainly cytokines, are directly involved in the immune system. Biological therapy has found a niche in the management of cancer, autoimmune diseases, and diseases of unknown cause that result in symptoms due to immune related mechanisms.

Bruton's tyrosine kinase, also known as tyrosine-protein kinase BTK, is a tyrosine kinase that is encoded by the BTK gene in humans. BTK plays a crucial role in B cell development.

Monoclonal antibody therapy is a form of immunotherapy that uses monoclonal antibodies (mAbs) to bind monospecifically to certain cells or proteins. The objective is that this treatment will stimulate the patient's immune system to attack those cells. Alternatively, in radioimmunotherapy a radioactive dose localizes a target cell line, delivering lethal chemical doses. Antibodies have been used to bind to molecules involved in T-cell regulation to remove inhibitory pathways that block T-cell responses. This is known as immune checkpoint therapy.
Galiximab is a monoclonal antibody designed for the treatment of B-cell lymphoma. As of September 2009, it is undergoing Phase III clinical trials. The drug is a chimeric antibody from Macaca irus and Homo sapiens.
Visilizumab is a humanized monoclonal antibody. It is being investigated for use as an immunosuppressive drug in patients with ulcerative colitis and Crohn's disease. Visilizumab binds to the CD3 receptor on certain activated T cells without affecting resting T cells. It is currently under clinical studies for the treatment of ulcerative colitis and Crohn's disease.
Priliximab is a human-mouse chimeric anti-CD4 monoclonal antibody. It has been tested on patients with Crohn's disease and multiple sclerosis but has not yet received U.S. Food and Drug Administration licensing. The patent belongs to the biotechnology company Centocor.

Interleukin-2 receptor alpha chain is a protein involved in assembly of high-affinity Interleukin-2 receptor, consisting of alpha (IL2RA), beta (IL2RB) and the common gamma chain (IL2RG). As the name indicates, this receptor interacts with pleiotropic cytokine called Interleukin-2, which effect is mainly important for immune homeostasis.
Short Course Immune Induction Therapy or SCIIT, is a therapeutic strategy employing rapid, specific, short term-modulation of the immune system using a therapeutic agent to induce T-cell non-responsiveness, also known as operational tolerance. As an alternative strategy to immunosuppression and antigen-specific tolerance inducing therapies, the primary goal of SCIIT is to re-establish or induce peripheral immune tolerance in the context of autoimmune disease and transplant rejection through the use of biological agents. In recent years, SCIIT has received increasing attention in clinical and research settings as an alternative to immunosuppressive drugs currently used in the clinic, drugs which put the patients at risk of developing infection, cancer, and cardiovascular disease.
David G. Maloney is an oncologist and researcher at Fred Hutchinson Cancer Research Center and the University of Washington who specializes in developing targeted immunotherapies for the treatment of blood cancers.
Otilimab is a fully human antibody which has been developed by the biotechnology company MorphoSys. It can also be referred to as HuCAL antibody, HuCAL standing for Human Combinatorial Antibody Library and being a technology used to generate monoclonal antibodies. Otilimab is directed against the granulocyte-macrophage colony stimulating factor (GM-CSF), a monomeric glycoprotein functioning as a cytokine promoting both proliferation and activation of macrophages and neutrophils.









