Expression
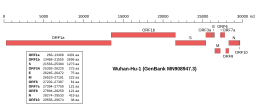 Genomic organisation of isolate Wuhan-Hu-1, the earliest sequenced sample of SARS-CoV-2, indicating the location of ORF1a and ORF1b | |
| NCBI genome ID | 86693 |
|---|---|
| Genome size | 29,903 bases |
| Year of completion | 2020 |
| Genome browser (UCSC) | |
ORF1a is the first open reading frame at the 5' end of the genome. Together ORF1ab occupies about two thirds of the genome, with the remaining third at the 3' end encoding the structural proteins and accessory proteins. [1] [2] [3] It is translated from a 5' capped RNA by cap-dependent translation. [1] Nidoviruses have a complex system of discontinuous subgenomic RNA production to enable expression of genes in their relatively large RNA genomes (typically 27-32kb for coronaviruses [1] ), but ORF1ab is translated directly from the genomic RNA. [5] ORF1ab sequences have been observed in noncanonical subgenomic RNAs, though their functional significance is unclear. [5]
A programmed ribosomal frameshift allows reading through the stop codon that terminates ORF1a to continue in a -1 reading frame, producing the longer polyprotein pp1ab. The frameshift occurs at a slippery sequence which is followed by a pseudoknot RNA secondary structure. [1] This has been measured at between 20-50% efficiency for murine coronavirus, [6] or 45-70% in SARS-CoV-2 [7] yielding a stoichiometry of roughly 1.5 to 2 times as much pp1a as pp1ab protein expressed. [2]


