
Bacillus cereus is a Gram-positive, rod-shaped, facultatively anaerobic, motile, beta-hemolytic, spore forming bacterium commonly found in soil and food. The specific name, cereus, meaning "waxy" in Latin, refers to the appearance of colonies grown on blood agar. Some strains are harmful to humans and cause foodborne illness, while other strains can be beneficial as probiotics for animals. The bacteria is classically contracted from fried rice dishes that have been sitting at room temperature for hours. B. cereus bacteria are facultative anaerobes, and like other members of the genus Bacillus, can produce protective endospores. Its virulence factors include cereolysin and phospholipase C.
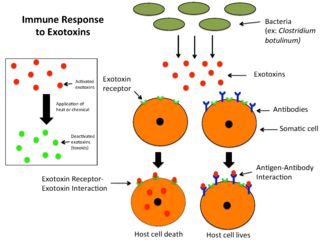
An exotoxin is a toxin secreted by bacteria. An exotoxin can cause damage to the host by destroying cells or disrupting normal cellular metabolism. They are highly potent and can cause major damage to the host. Exotoxins may be secreted, or, similar to endotoxins, may be released during lysis of the cell. Gram negative pathogens may secrete outer membrane vesicles containing lipopolysaccharide endotoxin and some virulence proteins in the bounding membrane along with some other toxins as intra-vesicular contents, thus adding a previously unforeseen dimension to the well-known eukaryote process of membrane vesicle trafficking, which is quite active at the host-pathogen interface.

An enterotoxin is a protein exotoxin released by a microorganism that targets the intestines.
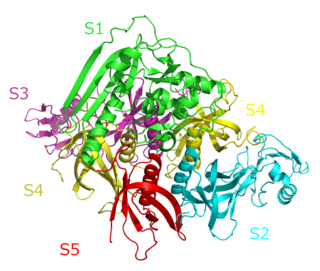
Pertussis toxin (PT) is a protein-based AB5-type exotoxin produced by the bacterium Bordetella pertussis, which causes whooping cough. PT is involved in the colonization of the respiratory tract and the establishment of infection. Research suggests PT may have a therapeutic role in treating a number of common human ailments, including hypertension, viral infection, and autoimmunity.
Collagenases are enzymes that break the peptide bonds in collagen. They assist in destroying extracellular structures in the pathogenesis of bacteria such as Clostridium. They are considered a virulence factor, facilitating the spread of gas gangrene. They normally target the connective tissue in muscle cells and other body organs.

Sphingomyelin phosphodiesterase is a hydrolase enzyme that is involved in sphingolipid metabolism reactions. SMase is a member of the DNase I superfamily of enzymes and is responsible for breaking sphingomyelin (SM) down into phosphocholine and ceramide. The activation of SMase has been suggested as a major route for the production of ceramide in response to cellular stresses.
Lecithinase is a type of phospholipase that acts upon lecithin.
Cytolysin refers to the substance secreted by microorganisms, plants or animals that is specifically toxic to individual cells, in many cases causing their dissolution through lysis. Cytolysins that have a specific action for certain cells are named accordingly. For instance, the cytolysins responsible for the destruction of red blood cells, thereby liberating hemoglobins, are named hemolysins, and so on. Cytolysins may be involved in immunity as well as in venoms.

Clostridium perfringens alpha toxin is a toxin produced by the bacterium Clostridium perfringens and is responsible for gas gangrene and myonecrosis in infected tissues. The toxin also possesses hemolytic activity.

Pore-forming proteins are usually produced by bacteria, and include a number of protein exotoxins but may also be produced by other organisms such as earthworms, who produce lysenin. They are frequently cytotoxic, as they create unregulated pores in the membrane of targeted cells.
Listeriolysin O (LLO) is a hemolysin produced by the bacterium Listeria monocytogenes, the pathogen responsible for causing listeriosis. The toxin may be considered a virulence factor, since it is crucial for the virulence of L. monocytogenes.

Phospholipase C (PLC) is a class of membrane-associated enzymes that cleave phospholipids just before the phosphate group (see figure). It is most commonly taken to be synonymous with the human forms of this enzyme, which play an important role in eukaryotic cell physiology, in particular signal transduction pathways. There are thirteen kinds of mammalian phospholipase C that are classified into six isotypes (β, γ, δ, ε, ζ, η) according to structure. Each PLC has unique and overlapping controls over expression and subcellular distribution. Activators of each PLC vary, but typically include heterotrimeric G protein subunits, protein tyrosine kinases, small G proteins, Ca2+, and phospholipids.
Clostridium enterotoxins are toxins produced by Clostridium species.

Clostridium difficile toxin A (TcdA) is a toxin generated by Clostridioides difficile, formerly known as Clostridium difficile. It is similar to Clostridium difficile Toxin B. The toxins are the main virulence factors produced by the gram positive, anaerobic, Clostridioides difficile bacteria. The toxins function by damaging the intestinal mucosa and cause the symptoms of C. difficile infection, including pseudomembranous colitis.

The AB toxins are two-component protein complexes secreted by a number of pathogenic bacteria. They can be classified as Type III toxins because they interfere with internal cell function. They are named AB toxins due to their components: the "A" component is usually the "active" portion, and the "B" component is usually the "binding" portion. The "A" subunit possesses enzyme activity, and is transferred to the host cell following a conformational change in the membrane-bound transport "B" subunit. These proteins consist of two independent polypeptides, which correspond to the A/B subunit moieties. The enzyme component (A) enters the cell through endosomes produced by the oligomeric binding/translocation protein (B), and prevents actin polymerisation through ADP-ribosylation of monomeric G-actin.
Clostridium novyi (oedematiens) a Gram-positive, endospore- forming, obligate anaerobic bacteria of the class clostridia. It is ubiquitous, being found in the soil and faeces. It is pathogenic, causing a wide variety of diseases in man and animals. It comes in three types, labelled A, B, and a non-pathogenic type C distinguished by the range of toxins they produce. Some authors include Clostridium haemolyticum as Clostridium novyi type D. C novyi is closely related to Clostridium botulinum types C and D as Yoshimasa Sasaki et al. have demonstrated by 16S rDNA sequence analysis.
Clostridium perfringens beta toxin is one of the four major lethal toxins produced by Clostridium perfringens Type B and Type C strains. It is a necrotizing agent and it induces hypertension by release of catecholamine. It has been shown to cause necrotic enteritis in mammals and induces necrotizing intestinal lesions in the rabbit ileal loop model. C. perfringens beta toxin is susceptible to breakdown by proteolytic enzymes, particularly trypsin. Beta toxin is therefore highly lethal to infant mammals because of trypsin inhibitors present in the colostrum.
Clostridium histolyticum is a species of bacteria found in feces and the soil. It is a motile, gram-positive, aerotolerant anaerobe. C. histolyticum is pathogenic in many species, including guinea pigs, mice, and rabbits, and humans. C. histolyticum has been shown to cause gas gangrene, often in association with other bacteria species.
The Thiol-activated Cholesterol-dependent Cytolysin(CDC) family is a member of the MACPF superfamily. Cholesterol dependent cytolysins are a family of β-barrel pore-forming exotoxins that are secreted by gram-positive bacteria. CDCs are secreted as water-soluble monomers of 50-70 kDa, that when bound to the target cell, form a circular homo-oligomeric complex containing as many as 40 monomers. Through multiple conformational changes, the β-barrel transmembrane structure is formed and inserted into the target cell membrane. The presence of cholesterol in the target membrane is required for pore formation, though the presence of cholesterol is not required by all CDCs for binding. For example, Intermedilysin secreted by Streptococcus intermedius will bind only to target membranes containing a specific protein receptor, independent of the presence of cholesterol, but cholesterol is required by intermedilysin for pore formation. While the lipid environment of cholesterol in the membrane can affect toxin binding, the exact molecular mechanism that cholesterol regulates the cytolytic activity of the CDC is not fully understood.
Phospholipase C (EC 3.1.4.3, lipophosphodiesterase I, Clostridium welchii alpha-toxin, Clostridium oedematiens beta- and gamma-toxins, lipophosphodiesterase C, phosphatidase C, heat-labile hemolysin, alpha-toxin) is an enzyme with systematic name phosphatidylcholine cholinephosphohydrolase. This enzyme catalyses the following chemical reaction











