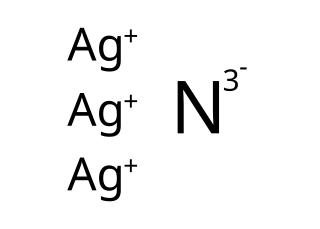Boron nitride is a thermally and chemically resistant refractory compound of boron and nitrogen with the chemical formula BN. It exists in various crystalline forms that are isoelectronic to a similarly structured carbon lattice. The hexagonal form corresponding to graphite is the most stable and soft among BN polymorphs, and is therefore used as a lubricant and an additive to cosmetic products. The cubic variety analogous to diamond is called c-BN; it is softer than diamond, but its thermal and chemical stability is superior. The rare wurtzite BN modification is similar to lonsdaleite but slightly softer than the cubic form.

Nitrogen is a chemical element; it has symbol N and atomic number 7. Nitrogen is a nonmetal and the lightest member of group 15 of the periodic table, often called the pnictogens. It is a common element in the universe, estimated at seventh in total abundance in the Milky Way and the Solar System. At standard temperature and pressure, two atoms of the element bond to form N2, a colorless and odorless diatomic gas. N2 forms about 78% of Earth's atmosphere, making it the most abundant uncombined element in air. Because of the volatility of nitrogen compounds, nitrogen is relatively rare in the solid parts of the Earth.

In chemistry, melon is a compound of carbon, nitrogen, and hydrogen of still somewhat uncertain composition, consisting mostly of heptazine units linked and closed by amine groups and bridges. It is a pale yellow solid, insoluble in most solvents.

Thallium(I) oxide is the inorganic compound of thallium and oxygen with the formula Tl2O in which thallium is in its +1 oxidation state. It is black and produces a basic yellow solution of thallium(I) hydroxide (TlOH) when dissolved in water. It is formed by heating solid TlOH or Tl2CO3 in the absence of air. Thallium oxide is used to make special high refractive index glass. Thallium oxide is a component of several high temperature superconductors. Thallium(I) oxide reacts with acids to make thallium(I) salts.
Lithium carbide, Li
2C
2, often known as dilithium acetylide, is a chemical compound of lithium and carbon, an acetylide. It is an intermediate compound produced during radiocarbon dating procedures. Li
2C
2 is one of an extensive range of lithium-carbon compounds which include the lithium-rich Li
4C, Li
6C
2, Li
8C
3, Li
6C
3, Li
4C
3, Li
4C
5, and the graphite intercalation compounds LiC
6, LiC
12, and LiC
18.
Li
2C
2 is the most thermodynamically-stable lithium-rich carbide and the only one that can be obtained directly from the elements. It was first produced by Moissan, in 1896 who reacted coal with lithium carbonate.
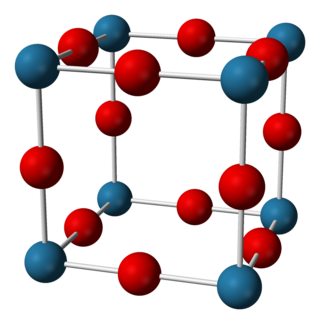
Sodium nitride is the inorganic compound with the chemical formula Na3N. In contrast to lithium nitride and some other nitrides, sodium nitride is an extremely unstable alkali metal nitride. It can be generated by combining atomic beams of sodium and nitrogen deposited onto a low-temperature sapphire substrate. It readily decomposes into its elements:

In organic chemistry, ethenone is the formal name for ketene, an organic compound with formula C2H2O or H2C=C=O. It is the simplest member of the ketene class. It is an important reagent for acetylations.
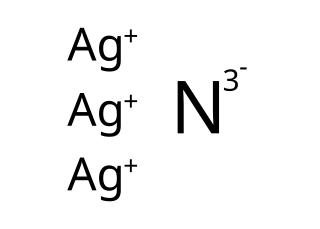
Silver nitride is an explosive chemical compound with symbol Ag3N. It is a black, metallic-looking solid which is formed when silver oxide or silver nitrate is dissolved in concentrated solutions of ammonia, causing formation of the diammine silver complex which subsequently breaks down to Ag3N. The standard free energy of the compound is about +315 kJ/mol, making it an endothermic compound which decomposes explosively to metallic silver and nitrogen gas.
Cadmium hydride is an inorganic compound with the chemical formula (CdH
2)
n. It is a solid, known only as a thermally unstable, insoluble white powder.

Tetrasilane is a silane with the structure formula SiH3–(SiH2)2–SiH3. It is the silane analog of butane.
Iron(II) iodide is an inorganic compound with the chemical formula FeI2. It is used as a catalyst in organic reactions.
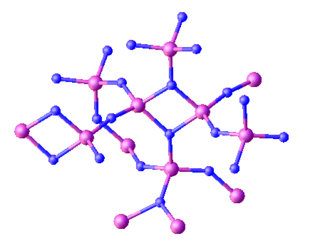
Triphosphorus pentanitride is an inorganic compound with the chemical formula P3N5. Containing only phosphorus and nitrogen, this material is classified as a binary nitride. While it has been investigated for various applications this has not led to any significant industrial uses. It is a white solid, although samples often appear colored owing to impurities.

Hydromelonic acid, is an elusive chemical compound with formula C
9H
3N
13 or (HNCN)
3(C
6N
7), whose molecule would consist of a heptazine H3(C
6N
7) molecule, with three cyanamido groups H–N=C=N– or N≡C–NH– substituted for the hydrogen atoms.
The inorganic imides are compounds containing an ion composed of nitrogen bonded to hydrogen with formula HN2−. Organic imides have the NH group, and two single or one double covalent bond to other atoms. The imides are related to the inorganic amides (H2N−), the nitrides (N3−) and the nitridohydrides (N3−•H−).
Potassium nitride is an unstable chemical compound. Several syntheses were erroneously claimed in the 19th century, and by 1894 it was assumed that it did not exist.
A chloride nitride is a mixed anion compound containing both chloride (Cl−) and nitride ions (N3−). Another name is metallochloronitrides. They are a subclass of halide nitrides or pnictide halides.
An iodide nitride is a mixed anion compound containing both iodide (I−) and nitride ions (N3−). Another name is metalloiodonitrides. They are a subclass of halide nitrides or pnictide halides. Some different kinds include ionic alkali or alkaline earth salts, small clusters where metal atoms surround a nitrogen atom, layered group 4 element 2-dimensional structures, and transition metal nitrido complexes counter-balanced with iodide ions. There is also a family with rare earth elements and nitrogen and sulfur in a cluster.

Europium compounds are compounds formed by the lanthanide metal europium (Eu). In these compounds, europium generally exhibits the +3 oxidation state, such as EuCl3, Eu(NO3)3 and Eu(CH3COO)3. Compounds with europium in the +2 oxidation state are also known. The +2 ion of europium is the most stable divalent ion of lanthanide metals in aqueous solution. Many europium compounds fluoresce under ultraviolet light due to the excitation of electrons to higher energy levels. Lipophilic europium complexes often feature acetylacetonate-like ligands, e.g., Eufod.
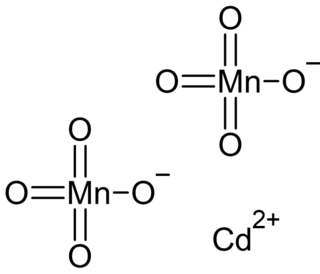
Cadmium permanganate is an inorganic compound with the chemical formula Cd(MnO4)2. It can form the hexahydrate Cd(MnO4)2·6H2O.
Americium nitride is a binary inorganic compound of americium and nitride with the chemical formula AmN.