 | |
| Identifiers | |
|---|---|
| |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.033.532 |
| EC Number |
|
PubChem CID | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| EuO4P | |
| Molar mass | 246.934 g·mol−1 |
| Appearance | colourless solid [1] |
| Density | 5.81 g·cm−3 [2] |
| Melting point | 2,200 °C (3,990 °F; 2,470 K) [3] |
| insoluble [4] | |
| Structure [2] | |
| Monazite | |
| P21/n (No. 14) | |
a = 668.13(10), b = 686.18(9), c = 634.91(8) pm α = 90°, β = 103.96(1)°, γ = 90° | |
| Thermochemistry | |
Heat capacity (C) | 111.5 J/mol·K [5] [6] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
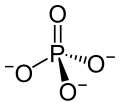
Europium(III) phosphate is one of the phosphates of europium, with the chemical formula of EuPO4. Other phosphates include europium(II) phosphate (Eu3(PO4)2) and europium(II,III) phosphate (Eu3Eu(PO4)3). [7]