
Manganese dioxide is the inorganic compound with the formula MnO
2. This blackish or brown solid occurs naturally as the mineral pyrolusite, which is the main ore of manganese and a component of manganese nodules. The principal use for MnO
2 is for dry-cell batteries, such as the alkaline battery and the zinc–carbon battery. MnO
2 is also used as a pigment and as a precursor to other manganese compounds, such as KMnO
4. It is used as a reagent in organic synthesis, for example, for the oxidation of allylic alcohols. MnO
2 has an α-polymorph that can incorporate a variety of atoms in the "tunnels" or "channels" between the manganese oxide octahedra. There is considerable interest in α-MnO
2 as a possible cathode for lithium-ion batteries.

Potassium permanganate is an inorganic compound with the chemical formula KMnO4. It is a purplish-black crystalline salt, which dissolves in water as K+ and MnO−
4 ions to give an intensely pink to purple solution.
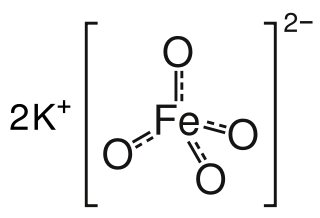
Potassium ferrate is an inorganic compound with the formula K2FeO4. It is the potassium salt of ferric acid. Potassium ferrate is a powerful oxidizing agent with applications in green chemistry, organic synthesis, and cathode technology.

A permanganate is a chemical compound with the manganate(VII) ion, MnO−
4, the conjugate base of permanganic acid. Because the manganese atom has a +7 oxidation state, the permanganate(VII) ion is a strong oxidising agent. The ion is a transition metal ion with a tetrahedral structure. Permanganate solutions are purple in colour and are stable in neutral or slightly alkaline media.

Sodium perchlorate is an inorganic compound with the chemical formula NaClO4. It consists of sodium cations Na+ and perchlorate anions ClO−4. It is a white crystalline, hygroscopic solid that is highly soluble in water and ethanol. It is usually encountered as sodium perchlorate monohydrate NaClO4·H2O. The compound is noteworthy as the most water-soluble of the common perchlorate salts.

In inorganic nomenclature, a manganate is any negatively charged molecular entity with manganese as the central atom. However, the name is usually used to refer to the tetraoxidomanganate(2−) anion, MnO2−
4, also known as manganate(VI) because it contains manganese in the +6 oxidation state. Manganates are the only known manganese(VI) compounds.

Sodium manganate is the inorganic compound with the formula Na2MnO4. This deep green solid is a rarely encountered analogue of the related salt K2MnO4. Sodium manganate is rare because it cannot be readily prepared from the oxidation of manganese dioxide and sodium hydroxide. Instead this oxidation reaction tends to stop at producing sodium hypomanganate, Na3MnO4, and even this Mn(V) salt is unstable in solution. Sodium manganate can be produced by reduction of sodium permanganate under basic conditions:
Permanganic acid (or manganic(VII) acid) is the inorganic compound with the formula HMnO4 and various hydrates. This strong oxoacid has been isolated as its dihydrate. It is the conjugate acid of permanganate salts. It is the subject of few publications and its characterization as well as its uses are very limited.

Sodium oxalate, or disodium oxalate, is a chemical compound with the chemical formula Na2C2O4. It is the sodium salt of oxalic acid. It contains sodium cations Na+ and oxalate anions C2O2−4. It is a white, crystalline, odorless solid, that decomposes above 290 °C.
Potassium manganate is the inorganic compound with the formula K2MnO4. This green-colored salt is an intermediate in the industrial synthesis of potassium permanganate, a common chemical. Occasionally, potassium manganate and potassium permanganate are confused, but each compound's properties are distinct.
In chemistry, hypomanganate, also called manganate(V) or tetraoxidomanganate(3−), is a trivalent anion (negative ion) composed of manganese and oxygen, with formula MnO3−
4.
Potassium hypomanganate is the inorganic compound with the formula K3MnO4. Also known as potassium manganate(V), this bright blue solid is a rare example of a salt with the hypomanganate or manganate(V) anion, where the manganese atom is in the +5 oxidation state. It is an intermediate in the production of potassium permanganate and the industrially most important Mn(V) compound.

Manganese(II) sulfate usually refers to the inorganic compound with the formula MnSO4·H2O. This pale pink deliquescent solid is a commercially significant manganese(II) salt. Approximately 260,000 tonnes of manganese(II) sulfate were produced worldwide in 2005. It is the precursor to manganese metal and many other chemical compounds. Manganese-deficient soil is remediated with this salt.
Permanganometry is one of the techniques used in chemical quantitative analysis. It is a redox titration that involves the use of permanganates to measure the amount of analyte present in unknown chemical samples. It involves two steps, namely the titration of the analyte with potassium permanganate solution and then the standardization of potassium permanganate solution with standard sodium oxalate solution. The titration involves volumetric manipulations to prepare the analyte solutions.

Ammonium permanganate is the chemical compound NH4MnO4, or NH3·HMnO4. It is a water soluble, violet-brown or dark purple salt.
In situ chemical oxidation (ISCO), a form of advanced oxidation process, is an environmental remediation technique used for soil and/or groundwater remediation to lower the concentrations of targeted environmental contaminants to acceptable levels. ISCO is accomplished by introducing strong chemical oxidizers into the contaminated medium to destroy chemical contaminants in place. It can be used to remediate a variety of organic compounds, including some that are resistant to natural degradation. The in situ in ISCO is just Latin for "in place", signifying that ISCO is a chemical oxidation reaction that occurs at the site of the contamination.

Manganese(II) molybdate is an inorganic compound with the chemical formula MnMoO4. α-MnMoO4 has a monoclinic crystal structure. It is also antiferromagnetic at low temperatures.
Barium permanganate is a chemical compound, with the formula Ba(MnO4)2. It forms violet to brown crystals that are sparingly soluble in water.

The chemical chameleon is a redox reaction, well known from classroom demonstrations, that exploits the dramatic color changes associated with the various oxidation states of manganese.
Rubidium permanganate is the permanganate salt of rubidium, with the chemical formula RbMnO
4.















