
5-HT receptors, 5-hydroxytryptamine receptors, or serotonin receptors, are a group of G protein-coupled receptor and ligand-gated ion channels found in the central and peripheral nervous systems. They mediate both excitatory and inhibitory neurotransmission. The serotonin receptors are activated by the neurotransmitter serotonin, which acts as their natural ligand.

SKF-82,958 is a synthetic compound of the benzazepine class that acts as a D1/D5 receptor full agonist. SKF-82,958 and similar D1-like-selective full agonists like SKF-81,297 and 6-Br-APB produce characteristic anorectic effects, hyperactivity and self-administration in animals, with a similar but not identical profile to that of dopaminergic stimulants such as amphetamine. SKF-82,958 was also subsequently found to act as an agonist of ERα with negligible activity at ERβ, making it a subtype-selective estrogen.

Pramipexole, sold under the brand Mirapex among others, is a medication used to treat Parkinson's disease (PD) and restless legs syndrome (RLS). In Parkinson's disease it may be used alone or together with levodopa. It is taken by mouth. Pramipexole is a dopamine agonist of the non-ergoline class.
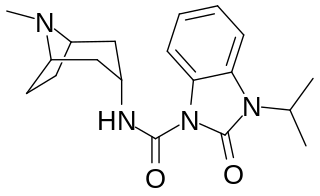
BIMU-8 is a drug which acts as a 5-HT4 receptor selective agonist. BIMU-8 was one of the first compounds of this class. The main action of BIMU-8 is to increase the rate of respiration by activating an area of the brain stem known as the pre-Botzinger complex.
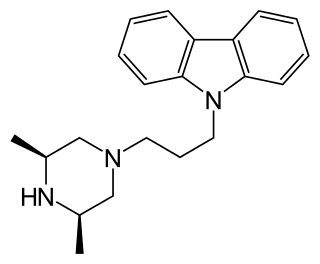
Rimcazole is an antagonist of the sigma receptor as well as a dopamine reuptake inhibitor. Sigma receptors are thought to be involved in the drug psychosis that can be induced by some drugs such as phencyclidine and cocaine, and rimcazole was originally researched as a potential antipsychotic with a different mechanism of action to traditional antipsychotic drugs. Trials proved inconclusive and rimcazole was not pursued for this application, but other sigma antagonists continue to be researched for a variety of potential applications. Rimcazole has been shown to reduce the effects of cocaine, and analogues of rimcazole have been shown to be highly effective at blocking the convulsions caused by cocaine overdose in animal models. Isothiocyanate derivatives of rimcazole acting as irreversible dopamine transporter (DAT) blockers have been developed.

SB-277,011A is a drug which acts as a potent and selective dopamine D3 receptor antagonist, which is around 80–100 times selective for D3 over D2, and lacks any partial agonist activity.

The serotonin 1A receptor is a subtype of serotonin receptors, or 5-HT receptors, that binds serotonin, also known as 5-HT, a neurotransmitter. 5-HT1A is expressed in the brain, spleen, and neonatal kidney. It is a G protein-coupled receptor (GPCR), coupled to the Gi protein, and its activation in the brain mediates hyperpolarization and reduction of firing rate of the postsynaptic neuron. In humans, the serotonin 1A receptor is encoded by the HTR1A gene.

Dopamine receptor D3 is a protein that in humans is encoded by the DRD3 gene.

WAY-100635 is a piperazine drug and research chemical widely used in scientific studies. It was originally believed to act as a selective 5-HT1A receptor antagonist, but subsequent research showed that it also acts as potent full agonist at the D4 receptor. It is sometimes referred to as a silent antagonist at the former receptor. It is closely related to WAY-100135.

N-n-Propylnorapomorphine (NPA) is an aporphine derivative dopamine agonist closely related to apomorphine. In rodents it has been shown to produce hyperactivity, stereotypy, hypothermia, antinociception, and penile erection, among other effects. Notably, its effects on locomotion are biphasic, with low doses producing inhibition and catalepsy and high doses resulting in enhancement of activity. This is likely due to preferential activation of D2/D3 autoreceptors versus postsynaptic receptors, the latter of which overcomes the former to increase postsynaptic dopaminergic signaling only with high doses.

Nemonapride, also previously known as emonapride and sold under the brand name Emilace, is an atypical antipsychotic which is used in the treatment of schizophrenia. It is taken by mouth.

UH-232 ((+)-UH232) is a drug which acts as a subtype selective mixed agonist-antagonist for dopamine receptors, acting as a weak partial agonist at the D3 subtype, and an antagonist at D2Sh autoreceptors on dopaminergic nerve terminals. It causes dopamine release in the brain and has a stimulant effect, as well as blocking the behavioural effects of cocaine. It may also serve as a 5-HT2A receptor agonist, based on animal studies. It was investigated in clinical trials for the treatment of schizophrenia, but unexpectedly caused symptoms to become worse.

A-68930 is a synthetic compound that acts as a selective dopamine receptor D1 agonist. It is orally active and has antidepressant and anorectic effects in animals, producing wakefulness and tachycardia, but without stimulant effects, instead producing sedation. The difference in effects between A-68930 and other D1 agonists such as SKF-82958 may be due to their differing effects on the related D5 receptor.

7-OH-DPAT is a synthetic compound that acts as a dopamine receptor agonist with reasonable selectivity for the D3 receptor subtype, and low affinity for serotonin receptors, unlike its structural isomer 8-OH-DPAT. 7-OH-DPAT is self-administered in several animal models, and is used to study its addiction effects to cocaine.
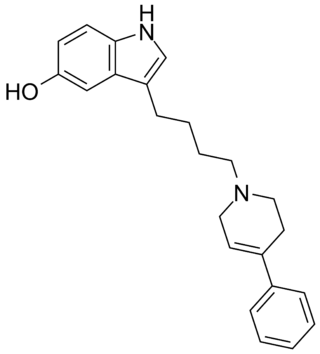
Roxindole (EMD-49,980) is a dopaminergic and serotonergic drug which was originally developed by Merck KGaA for the treatment of schizophrenia. In clinical trials its antipsychotic efficacy was only modest but it was unexpectedly found to produce potent and rapid antidepressant and anxiolytic effects. As a result, roxindole was further researched for the treatment of depression instead. It has also been investigated as a therapy for Parkinson's disease and prolactinoma.
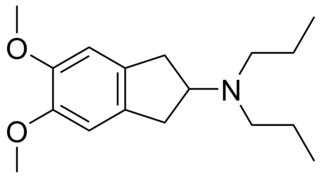
PNU-99,194(A) (or U-99,194(A)) is a drug which acts as a moderately selective D3 receptor antagonist with ~15-30-fold preference for D3 over the D2 subtype. Though it has substantially greater preference for D3 over D2, the latter receptor does still play some role in its effects, as evidenced by the fact that PNU-99,194 weakly stimulates both prolactin secretion and striatal dopamine synthesis, actions it does not share with the more selective (100-fold) D3 receptor antagonists S-14,297 and GR-103,691.

CY-208,243 is a drug which acts as a dopamine agonist selective for the D1 subtype. Unlike most D1-selective agonists, it shows efficacy in animal models of Parkinson's disease.

CP-93129 is a drug which acts as a potent and selective serotonin 5-HT1B receptor agonist, with approximately 150x and 200x selectivity over the closely related 5-HT1D and 5-HT1A receptors. It is used in the study of 5-HT1B receptors in the brain, particularly their role in modulating the release of other neurotransmitters.

L-741,626 is a drug which acts as a potent and selective antagonist for the dopamine receptor D2. It has good selectivity over the related D3 and D4 subtypes and other receptors. L-741,626 is used for laboratory research into brain function and has proved particularly useful for distinguishing D2 mediated responses from those produced by the closely related D3 subtype, and for studying the roles of these subtypes in the action of cocaine and amphetamines in the brain.

Bay R 1531 is a tricyclic tryptamine derivative which acts as a selective serotonin receptor 5-HT1A agonist. It was researched unsuccessfully for the treatment of stroke but remains in use for scientific research.




















