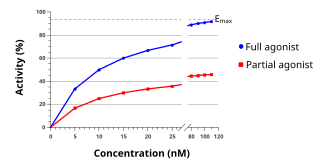
An agonist is a chemical that activates a receptor to produce a biological response. Receptors are cellular proteins whose activation causes the cell to modify what it is currently doing. In contrast, an antagonist blocks the action of the agonist, while an inverse agonist causes an action opposite to that of the agonist.

Azapirones are a class of drugs used as anxiolytics, antidepressants, and antipsychotics. They are commonly used as add-ons to other antidepressants, such as selective serotonin reuptake inhibitors (SSRIs).

Amoxapine, sold under the brand name Asendin among others, is a tricyclic antidepressant (TCAs). It is the N-demethylated metabolite of loxapine. Amoxapine first received marketing approval in the United States in 1992.

A dopamine antagonist, also known as an anti-dopaminergic and a dopamine receptor antagonist (DRA), is a type of drug which blocks dopamine receptors by receptor antagonism. Most antipsychotics are dopamine antagonists, and as such they have found use in treating schizophrenia, bipolar disorder, and stimulant psychosis. Several other dopamine antagonists are antiemetics used in the treatment of nausea and vomiting.

Tiotixene, or thiothixene, sold under the brand name Navane among others, is a typical antipsychotic of the thioxanthene class which is related to chlorprothixene and is used in the treatment of psychoses like schizophrenia and bipolar mania. It was introduced in the United States in 1967 by Pfizer.

Amisulpride is an antiemetic and antipsychotic medication used at lower doses intravenously to prevent and treat postoperative nausea and vomiting; and at higher doses by mouth to treat schizophrenia and acute psychotic episodes. It is sold under the brand names Barhemsys and Solian, Socian, Deniban and others. It is also used to treat dysthymia.

Zotepine is an atypical antipsychotic drug indicated for acute and chronic schizophrenia. It has been used in Germany since 1990 and Japan since 1982.

Dopamine receptor D1, also known as DRD1. It is one of the two types of D1-like receptor family - receptors D1 and D5. It is a protein that in humans is encoded by the DRD1 gene.
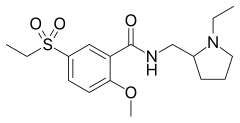
Tiapride is a drug that selectively blocks D2 and D3 dopamine receptors in the brain. It is used to treat a variety of neurological and psychiatric disorders including dyskinesia, alcohol withdrawal syndrome, negative symptoms of psychosis, and agitation and aggression in the elderly. A derivative of benzamide, tiapride is chemically and functionally similar to other benzamide antipsychotics such as sulpiride and amisulpride known for their dopamine antagonist effects.

LY-379,268 is a drug that is used in neuroscience research, which acts as a potent and selective agonist for the group II metabotropic glutamate receptors (mGluR2/3).
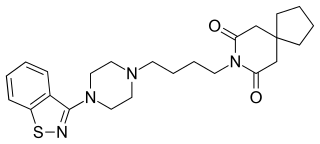
Tiospirone (BMY-13,859), also sometimes called tiaspirone or tiosperone, is an atypical antipsychotic of the azapirone class. It was investigated as a treatment for schizophrenia in the late 1980s and was found to have an effectiveness equivalent to those of typical antipsychotics in clinical trials but without causing extrapyramidal side effects. However, development was halted and it was not marketed. Perospirone, another azapirone derivative with antipsychotic properties, was synthesized and assayed several years after tiospirone. It was found to be both more potent and more selective in comparison and was commercialized instead.
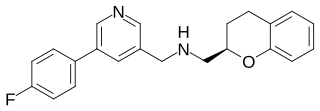
Sarizotan (EMD-128,130) is a selective 5-HT1A receptor agonist and D2 receptor antagonist, which has antipsychotic effects, and has also shown efficacy in reducing dyskinesias resulting from long-term anti-Parkinsonian treatment with levodopa.

N-Desmethylclozapine (NDMC), or norclozapine, is a major active metabolite of the atypical antipsychotic drug clozapine. Unlike clozapine, it possesses intrinsic activity at the D2/D3 receptors, and acts as a weak partial agonist at these sites similarly to aripiprazole and bifeprunox. Notably, NDMC has also been shown to act as a potent and efficacious agonist at the M1 and δ-opioid receptors, unlike clozapine as well. It was hypothesized that on account of these unique actions, NDMC might underlie the clinical superiority of clozapine over other antipsychotics. However, clinical trials found NMDC itself ineffective in the treatment of schizophrenia. This may be because it possesses relatively low D2/D3 occupancy compared to 5-HT2 (<15% versus 64-79% at a dose of 10–60 mg/kg s.c. in animal studies). Albeit not useful in the treatment of positive symptoms on its own, it cannot be ruled out that NDMC may contribute to the efficacy of clozapine on cognitive and/or negative symptoms.
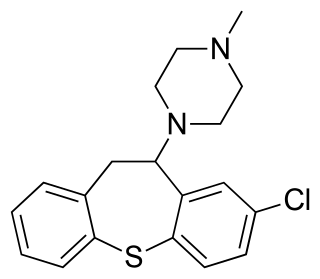
Clorotepine, also known as octoclothepin or octoclothepine, is an antipsychotic of the tricyclic group which was derived from perathiepin in 1965 and marketed in the Czech Republic by Spofa in or around 1971 for the treatment of schizophrenic psychosis.
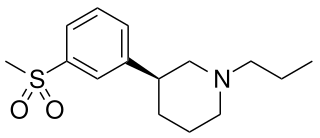
OSU-6162 (PNU-96391) is a compound which acts as a partial agonist at both dopamine D2 receptors and 5-HT2A receptors. It acts as a dopamine stabilizer in a similar manner to the closely related drug pridopidine, and has antipsychotic, anti-addictive and anti-Parkinsonian effects in animal studies. Both enantiomers show similar activity but with different ratios of effects, with the (S) enantiomer (–)-OSU-6162 that is more commonly used in research, having higher binding affinity to D2 but is a weaker partial agonist at 5-HT2A, while the (R) enantiomer (+)-OSU-6162 has higher efficacy at 5-HT2A but lower D2 affinity.

F-15,063 is an orally active potential antipsychotic, and an antagonist at the D2/D3 receptors, partial agonist at the D4 receptor, and agonist at the 5-HT1A receptors. It has greater efficacy at the 5-HT1A receptors than other antipsychotics, such as clozapine, aripiprazole, and ziprasidone. This greater efficacy may lead to enhanced antipsychotic properties, as antipsychotics that lack 5-HT1A affinity are associated with increased risk of extrapyramidal symptoms, and lack of activity against the negative symptoms of schizophrenia.

Brexpiprazole, sold under the brand name Rexulti among others, is an atypical antipsychotic. It is a dopamine D2 receptor partial agonist and has been described as a "serotonin–dopamine activity modulator" (SDAM). The drug was approved by the U.S. Food and Drug Administration (FDA) on July 10, 2015, for the treatment of schizophrenia, and as an adjunctive treatment for depression. It has been designed to provide improved efficacy and tolerability (e.g., less akathisia, restlessness and/or insomnia) over established adjunctive treatments for major depressive disorder (MDD).

Levofenfluramine (INN), or (−)-3-trifluoromethyl-N-ethylamphetamine, also known as (−)-fenfluramine or (R)-fenfluramine, is a drug of the amphetamine family that, itself, was never marketed. It is the levorotatory enantiomer of fenfluramine, the racemic form of the compound, whereas the dextrorotatory enantiomer is dexfenfluramine. Both fenfluramine and dexfenfluramine are anorectic agents that have been used clinically in the treatment of obesity. However, they have since been discontinued due to reports of causing cardiovascular conditions such as valvular heart disease and pulmonary hypertension, adverse effects that are likely to be caused by excessive stimulation of 5-HT2B receptors expressed on heart valves.

Acetothiolutamide is a selective androgen receptor modulator (SARM) derived from the nonsteroidal antiandrogen bicalutamide that was described in 2002 and was one of the first SARMs to be discovered and developed. It is a high-affinity, selective ligand of the androgen receptor (AR), where it acts as a full agonist in vitro, and has in vitro potency comparable to that of testosterone. However, in vivo, acetothiolutamide displayed overall negligible androgenic effects, though significant anabolic effects were observed at high doses. In addition, notable antiandrogen effects were observed in castrated male rats treated with testosterone propionate. The discrepancy between the in vitro and in vivo actions of acetothiolutamide was determined to be related to rapid plasma clearance and extensive hepatic metabolism into a variety of metabolites with differing pharmacological activity, including AR partial agonism and antagonism. In accordance with its poor metabolic stability, acetothiolutamide is not orally bioavailable, and shows activity only via injected routes such as subcutaneous and intravenous.

AC-90179 is a piperidine derivative which acts as an inverse agonist at the 5-HT2A serotonin receptor and an antagonist at 5-HT2C. It was developed as a potential antipsychotic but was not pursued for medical applications due to poor oral bioavailability, however it continues to be used as a tool compound in pharmacological research.