
Acetylcholine (ACh) is an organic compound that functions in the brain and body of many types of animals as a neurotransmitter. Its name is derived from its chemical structure: it is an ester of acetic acid and choline. Parts in the body that use or are affected by acetylcholine are referred to as cholinergic.

An acetylcholine receptor or a cholinergic receptor is an integral membrane protein that responds to the binding of acetylcholine, a neurotransmitter.

Muscarine, L-(+)-muscarine, or muscarin is a natural product found in certain mushrooms, particularly in Inocybe and Clitocybe species, such as the deadly C. dealbata. Mushrooms in the genera Entoloma and Mycena have also been found to contain levels of muscarine which can be dangerous if ingested. Muscarine has been found in harmless trace amounts in Boletus, Hygrocybe, Lactarius and Russula. Trace concentrations of muscarine are also found in Amanita muscaria, though the pharmacologically more relevant compound from this mushroom is the Z-drug-like alkaloid muscimol. A. muscaria fruitbodies contain a variable dose of muscarine, usually around 0.0003% fresh weight. This is very low and toxicity symptoms occur very rarely. Inocybe and Clitocybe contain muscarine concentrations up to 1.6%.
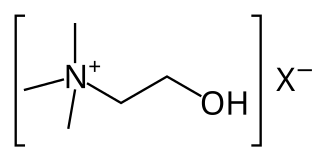
Cholinergic agents are compounds which mimic the action of acetylcholine and/or butyrylcholine. In general, the word "choline" describes the various quaternary ammonium salts containing the N,N,N-trimethylethanolammonium cation. Found in most animal tissues, choline is a primary component of the neurotransmitter acetylcholine and functions with inositol as a basic constituent of lecithin. Choline also prevents fat deposits in the liver and facilitates the movement of fats into cells.

The enzyme cholinesterase (EC 3.1.1.8, choline esterase; systematic name acylcholine acylhydrolase) catalyses the hydrolysis of choline-based esters:

A neuromuscular junction is a chemical synapse between a motor neuron and a muscle fiber.

Nicotinic acetylcholine receptors, or nAChRs, are receptor polypeptides that respond to the neurotransmitter acetylcholine. Nicotinic receptors also respond to drugs such as the agonist nicotine. They are found in the central and peripheral nervous system, muscle, and many other tissues of many organisms. At the neuromuscular junction they are the primary receptor in muscle for motor nerve-muscle communication that controls muscle contraction. In the peripheral nervous system: (1) they transmit outgoing signals from the presynaptic to the postsynaptic cells within the sympathetic and parasympathetic nervous system, and (2) they are the receptors found on skeletal muscle that receive acetylcholine released to signal for muscular contraction. In the immune system, nAChRs regulate inflammatory processes and signal through distinct intracellular pathways. In insects, the cholinergic system is limited to the central nervous system.

Muscarinic acetylcholine receptors, or mAChRs, are acetylcholine receptors that form G protein-coupled receptor complexes in the cell membranes of certain neurons and other cells. They play several roles, including acting as the main end-receptor stimulated by acetylcholine released from postganglionic fibers. They are mainly found in the parasympathetic nervous system, but also have a role in the sympathetic nervous system in the control of sweat glands.
Pseudocholinesterase deficiency is an autosomal recessive inherited blood plasma enzyme abnormality in which the body's production of butyrylcholinesterase is impaired. People who have this abnormality may be sensitive to certain anesthetic drugs, including the muscle relaxants succinylcholine and mivacurium as well as other ester local anesthetics.

Epibatidine is a chlorinated alkaloid that is secreted by the Ecuadoran frog Epipedobates anthonyi and poison dart frogs from the Ameerega genus. It was discovered by John W. Daly in 1974, but its structure was not fully elucidated until 1992. Whether epibatidine occurs naturally remains controversial due to challenges in conclusively identifying the compound from the limited samples collected by Daly. By the time that high-resolution spectrometry was used in 1991, there remained less than one milligram of extract from Daly's samples, raising concerns about possible contamination. Samples from other batches of the same species of frog failed to yield epibatidine.

Desformylflustrabromine (dFBr) is a NMT derivative indole alkaloid which was first isolated from the marine bryozoan Flustra foliacea.

The human muscarinic acetylcholine receptor M5, encoded by the CHRM5 gene, is a member of the G protein-coupled receptor superfamily of integral membrane proteins. It is coupled to Gq protein. Binding of the endogenous ligand acetylcholine to the M5 receptor triggers a number of cellular responses such as adenylate cyclase inhibition, phosphoinositide degradation, and potassium channel modulation. Muscarinic receptors mediate many of the effects of acetylcholine in the central and peripheral nervous system. The clinical implications of this receptor have not been fully explored; however, stimulation of this receptor is known to effectively decrease cyclic AMP levels and downregulate the activity of protein kinase A (PKA).

The muscarinic acetylcholine receptor, also known as cholinergic/acetylcholine receptor M3, or the muscarinic 3, is a muscarinic acetylcholine receptor encoded by the human gene CHRM3.

The muscarinic acetylcholine receptor M4, also known as the cholinergic receptor, muscarinic 4 (CHRM4), is a protein that, in humans, is encoded by the CHRM4 gene.

Tebanicline is a potent synthetic nicotinic (non-opioid) analgesic drug developed by Abbott. It was developed as a less toxic analog of the potent poison dart frog-derived compound epibatidine, which is about 200 times stronger than morphine as an analgesic, but produces extremely dangerous toxic side effects. Like epibatidine, tebanicline showed potent analgesic activity against neuropathic pain in both animal and human trials, but with far less toxicity than its parent compound. It acts as a partial agonist at neuronal nicotinic acetylcholine receptors, binding to both the α3β4 and the α4β2 subtypes.

AR-R17779 is a drug that acts as a potent and selective full agonist for the α7 subtype of neural nicotinic acetylcholine receptors. It has nootropic effects in animal studies, but its effects do not substitute for those of nicotine. It has also been studied as a potential novel treatment for arthritis.

Altinicline is a drug which acts as an agonist at neural nicotinic acetylcholine receptors with high selectivity for the α4β2 subtype. It stimulates release of dopamine and acetylcholine in the brain in both rodent and primate models, and progressed as far as Phase II clinical trials for Parkinson's disease, where "no antiparkinsonian or cognitive-enhancing effects were demonstrated", although its current status is unclear.

PNU-282,987 is a drug that acts as a potent and selective agonist for the α7 subtype of neural nicotinic acetylcholine receptors. In animal studies, it shows nootropic effects, and derivatives may be useful in the treatment of schizophrenia, although PNU-282,987 is not suitable for use in humans because of excessive inhibition of the hERG antitarget. PNU-282987 has been shown to initiate signaling that leads to adult neurogeneis in mammals.

Benzilylcholine mustard (N-2-chloroethyl-N-methyl 2-aminoethyl benzilate) is a modified version of acetylcholine, so named because after cyclization in solution it forms an aziridinium derivative that is structurally similar to benzilylcholine. It is well known for being an irreversible antagonist of the muscarinic acetylcholine receptor. It has been used in pharmacological experiments investigating the relationship between receptor occupancy and response. It was also one of the tools in characterization of the muscarinic acetylcholine receptor.

Neuromuscular drugs are chemical agents that are used to alter the transmission of nerve impulses to muscles, causing effects such as temporary paralysis of targeted skeletal muscles. Most neuromuscular drugs are available as quaternary ammonium compounds which are derived from acetylcholine (ACh). This allows neuromuscular drugs to act on multiple sites at neuromuscular junctions, mainly as antagonists or agonists of post-junctional nicotinic receptors. Neuromuscular drugs are classified into four main groups, depolarizing neuromuscular blockers, non-depolarizing neuromuscular blockers, acetylcholinesterase inhibitors, and butyrylcholinesterase inhibitors.


















