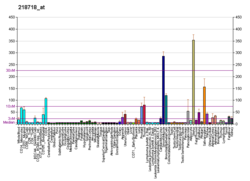
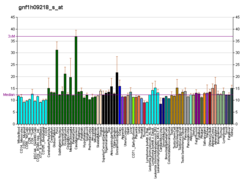
Platelet-derived growth factor C, also known as PDGF-C, is a 345-amino acid protein that in humans is encoded by the PDGFC gene. [5] [6] Platelet-derived growth factors are important in connective tissue growth, survival and function, and consist of disulphide-linked dimers involving two polypeptide chains, PDGF-A and PDGF-B. PDGF-C is a member of the PDGF/VEGF family of growth factors with a unique two-domain structure and expression pattern. PDGF-C was not previously identified with PDGF-A and PDGF-B, possibly because it may be that it is synthesized and secreted as a latent growth factor, requiring proteolytic removal of the N-terminal CUB domain for receptor binding and activation. [7]