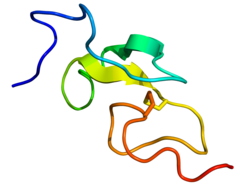
Epidermal growth factor (EGF) is a protein that stimulates cell growth and differentiation by binding to its receptor, EGFR. Human EGF is 6-kDa [5] and has 53 amino acid residues and three intramolecular disulfide bonds. [6]
Contents
- Structure
- Function
- Biological sources
- Polypeptide growth factors
- Mechanism
- EGF-family / EGF-like domain
- Interactions
- Medical uses
- Bone regeneration
- History
- References
- Further reading
- External links
EGF was originally described as a secreted peptide found in the submaxillary glands of mice and in human urine. EGF has since been found in many human tissues, including platelets, [7] submandibular gland (submaxillary gland), [8] and parotid gland. [8] Initially, human EGF was known as urogastrone. [9]