Related Research Articles

Gefitinib (ZD1839) is a drug used for certain breast, lung and other cancers. Gefitinib is an EGFR inhibitor, like erlotinib, which interrupts signaling through the epidermal growth factor receptor (EGFR) in target cells. Therefore, it is only effective in cancers with mutated and overactive EGFR. It is marketed by AstraZeneca and Teva.

Erlotinib, sold under the brand name Tarceva among others, is a medication used to treat non-small cell lung cancer (NSCLC) and pancreatic cancer. Specifically it is used for NSCLC with mutations in the epidermal growth factor receptor (EGFR) — either an exon 19 deletion (del19) or exon 21 (L858R) substitution mutation — which has spread to other parts of the body. It is taken by mouth.
Mapatumumab (HGS-ETR1) is an experimental human monoclonal antibody undergoing clinical trials for the treatment of cancer. It targets TRAIL-R1, also known as DR4, which is expressed on the surface of many tumor cell types.

Afatinib, sold under the brand name Gilotrif among others, is a medication used to treat non-small cell lung carcinoma (NSCLC). It belongs to the tyrosine kinase inhibitor family of medications. It is taken by mouth.
Ramucirumab is a fully human monoclonal antibody (IgG1) developed for the treatment of solid tumors. This drug was developed by ImClone Systems Inc. It was isolated from a native phage display library from Dyax.
Tigatuzumab (CS-1008) is a monoclonal antibody for the treatment of cancer. As of October 2009, a clinical trial for the treatment of pancreatic cancer, Phase II trials for colorectal cancer, non-small cell lung cancer, and ovarian cancer have been completed.
Figitumumab is a monoclonal antibody targeting the insulin-like growth factor-1 receptor that was investigated for the treatment of various types of cancer, for example adrenocortical carcinoma and non-small cell lung cancer (NSCLC).
Tecemotide is a synthetic lipopeptide that is used as antigen in an investigational therapeutic cancer vaccine. The investigational therapeutic cancer vaccine is designed to induce a cellular immune response to cancer cells that express MUC1, a glycoprotein antigen that is widely over-expressed on common cancers such as lung cancer, breast cancer, prostate cancer and colorectal cancer. The cellular immune response may lead then to a rejection of tumor tissue expressing the MUC1 antigen.
ALK inhibitors are anti-cancer drugs that act on tumours with variations of anaplastic lymphoma kinase (ALK) such as an EML4-ALK translocation.. They fall under the category of tyrosine kinase inhibitors, which work by inhibiting proteins involved in the abnormal growth of tumour cells. All the current approved ALK inhibitors function by binding to the ATP pocket of the abnormal ALK protein, blocking its access to energy and deactivating it. A majority of ALK-rearranged NSCLC harbour the EML4-ALK fusion, although as of 2020, over 92 fusion partners have been discovered in ALK+ NSCLC. For each fusion partner, there can be several fusion variants depending on the position the two genes were fused at, and this may have implications on the response of the tumour and prognosis of the patient.

Brigatinib, sold under the brand name Alunbrig among others, is a small-molecule targeted cancer therapy being developed by ARIAD Pharmaceuticals, Inc. Brigatinib acts as both an anaplastic lymphoma kinase (ALK) and epidermal growth factor receptor (EGFR) inhibitor.
Onartuzumab is a humanized monoclonal antibody designed for the treatment of advanced non-small-cell lung cancer.
Simtuzumab is a humanized monoclonal antibody designed for the treatment of fibrosis. It binds to LOXL2 and acts as an immunomodulator. In January 2016, Gilead Sciences terminated its Phase 2 clinical study in patients with idiopathic pulmonary fibrosis (IPF) due to lack of efficacy.
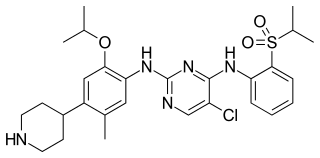
Ceritinib is a prescription-only drug used for the treatment of non-small cell lung cancer (NSCLC). It was developed by Novartis and received FDA approval for use in April 2014.

Encenicline is a selective partial agonist of the α7 nicotinic receptor, and is in phase III clinical trials for the treatment of cognitive impairment in schizophrenia.
Durvalumab is an FDA-approved immunotherapy for cancer, developed by Medimmune/AstraZeneca. It is a human immunoglobulin G1 kappa (IgG1κ) monoclonal antibody that blocks the interaction of programmed cell death ligand 1 (PD-L1) with the PD-1 (CD279).
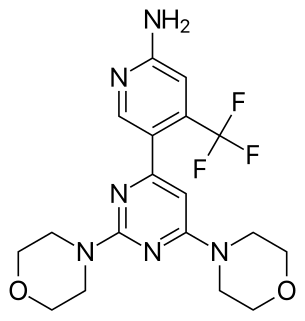
Buparlisib is an investigational small molecule orally-available pan-class I phosphoinositide 3-kinase inhibitor.

Alectinib is an oral drug that blocks the activity of anaplastic lymphoma kinase (ALK) and is used to treat non-small-cell lung cancer (NSCLC). It was developed by Chugai Pharmaceutical Co. Japan, which is part of the Hoffmann-La Roche group.
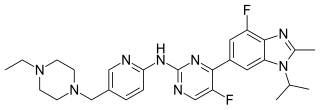
Abemaciclib is a drug for the treatment of advanced or metastatic breast cancers. It was developed by Eli Lilly and it acts as a CDK inhibitor selective for CDK4 and CDK6.
Entrectinib, sold under the brand name Rozlytrek, is an anti-cancer medication used to treat ROS1-positive non-small cell lung cancer and NTRK fusion-positive solid tumors. It is a selective tyrosine kinase inhibitor (TKI), of the tropomyosin receptor kinases (TRK) A, B and C, C-ros oncogene 1 (ROS1) and anaplastic lymphoma kinase (ALK).
Mirikizumab is a human monoclonal antibody designed for the treatment of psoriasis.
References
- ↑ World Health Organization (2014). "International Nonproprietary Names for Pharmaceutical Substances (INN). Proposed INN: List 111" (PDF). WHO Drug Information. 28 (2).
- ↑ Statement On A Nonproprietary Name Adopted By The USAN Council - Emibetuzumab, American Medical Association .
- ↑ Camidge DR, Moran T, Demedts I, Grosch H, Di Mercurio JP, Mileham KF, et al. "A randomized, open-label, phase 2 study of emibetuzumab plus erlotinib (LY+E) and emibetuzumab monotherapy (LY) in patients with acquired resistance to erlotinib and MET diagnostic positive (MET Dx+) metastatic NSCLC".
| This monoclonal antibody–related article is a stub. You can help Wikipedia by expanding it. |