
Separase, also known as separin, is a cysteine protease responsible for triggering anaphase by hydrolysing cohesin, which is the protein responsible for binding sister chromatids during the early stage of anaphase. In humans, separin is encoded by the ESPL1 gene.
SMC complexes represent a large family of ATPases that participate in many aspects of higher-order chromosome organization and dynamics. SMC stands for Structural Maintenance of Chromosomes.

Cohesin is a protein complex that mediates sister chromatid cohesion, homologous recombination, and DNA looping. Cohesin is formed of SMC3, SMC1, SCC1 and SCC3. Cohesin holds sister chromatids together after DNA replication until anaphase when removal of cohesin leads to separation of sister chromatids. The complex forms a ring-like structure and it is believed that sister chromatids are held together by entrapment inside the cohesin ring. Cohesin is a member of the SMC family of protein complexes which includes Condensin, MukBEF and SMC-ScpAB.
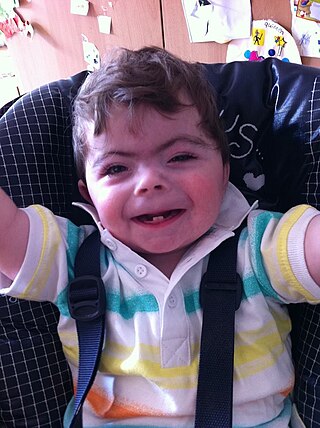
Cornelia de Lange syndrome (CdLS) is a genetic disorder. People with this syndrome experience a range of physical, cognitive, and medical challenges ranging from mild to severe. The syndrome has a widely varied phenotype, meaning people with the syndrome have varied features and challenges. The typical features of CdLS include thick or long eyebrows, a small nose, small stature, developmental delay, long or smooth philtrum, thin upper lip and downturned mouth.

Nipped-B-like protein (NIPBL), also known as SCC2 or delangin is a protein that in humans is encoded by the NIPBL gene. NIPBL is required for the association of cohesin with DNA and is the major subunit of the cohesin loading complex. Heterozygous mutations in NIPBL account for an estimated 60% of case of Cornelia de Lange Syndrome.

Structural maintenance of chromosomes protein 1A (SMC1A) is a protein that in humans is encoded by the SMC1A gene. SMC1A is a subunit of the cohesin complex which mediates sister chromatid cohesion, homologous recombination and DNA looping. In somatic cells, cohesin is formed of SMC1A, SMC3, RAD21 and either SA1 or SA2 whereas in meiosis, cohesin is formed of SMC3, SMC1B, REC8 and SA3.

Double-strand-break repair protein rad21 homolog is a protein that in humans is encoded by the RAD21 gene. RAD21, an essential gene, encodes a DNA double-strand break (DSB) repair protein that is evolutionarily conserved in all eukaryotes from budding yeast to humans. RAD21 protein is a structural component of the highly conserved cohesin complex consisting of RAD21, SMC1A, SMC3, and SCC3 [ STAG1 (SA1) and STAG2 (SA2) in multicellular organisms] proteins, involved in sister chromatid cohesion.

Sister chromatid cohesion protein DCC1 is a protein that in humans is encoded by the DSCC1 gene.

Cohesin subunit SA-2 (SA2) is a protein that in humans is encoded by the STAG2 gene. SA2 is a subunit of the Cohesin complex which mediates sister chromatid cohesion, homologous recombination and DNA looping. In somatic cells cohesin is formed of SMC3, SMC1, RAD21 and either SA1 or SA2 whereas in meiosis, cohesin is formed of SMC3, SMC1B, REC8 and SA3.

Wings apart-like protein homolog (WAPL) is a protein that in humans is encoded by the WAPAL gene. WAPL is a key regulator of the Cohesin complex which mediates sister chromatid cohesion, homologous recombination and DNA looping. Cohesin is formed of SMC3, SMC1, RAD21 and either SA1 or SA2. Cohesin has a ring-like arrangement and it is thought that it associates with the chromosome by entrapping it whether as a loop of DNA, a single strand or a pair of sister chromosomes. WAPL forms a complex with PDS5A or PDS5B and releases cohesin from DNA by opening the interface between SMC3 and RAD21.

Shugoshin 1 or Shugoshin-like 1, is a protein that in humans is encoded by the SGO1 gene.

Shugoshin 2(Shugoshin-2), also known as Shugoshin-like 2, is a protein which in humans is encoded by the SGO2 gene.

Sister chromatid cohesion protein PDS5 homolog B(PDS5B) is a protein that in humans is encoded by the PDS5B gene. It is a regulatory subunit of the Cohesin complex which mediates sister chromatid cohesion, homologous recombination and DNA looping. The core cohesin complex is formed of SMC3, SMC1, RAD21 and either SA1 or SA2. PDS5 associates with WAPL to stimulate the release of cohesin from DNA but during DNA replication PDS5 promotes acetylation of SMC3 by ESCO1 and ESCO2.

Meiotic recombination protein REC8 homolog is a protein that in humans is encoded by the REC8 gene.

Cohesin subunit SA-1 (SA1) is a protein that in humans is encoded by the STAG1 gene. SA1 is a subunit of the Cohesin complex which mediates sister chromatid cohesion, homologous recombination and DNA looping. In somatic cells cohesin is formed of SMC3, SMC1, RAD21 and either SA1 or SA2 whereas in meiosis, cohesin is formed of SMC3, SMC1B, REC8 and SA3. There is a nonprofit community formed for those with a STAG1 Gene mutation at www.stag1gene.org.

Structural maintenance of chromosomes protein 2 (SMC-2), also known as chromosome-associated protein E (CAP-E), is a protein that in humans is encoded by the SMC2 gene. SMC2 is part of the SMC protein family and is a core subunit of condensin I and II, large protein complexes involved in chromosome condensation, overall organization. Several studies have demonstrated the necessity of SMC2 for cell division and proliferation.

SLX4 is a protein involved in DNA repair, where it has important roles in the final steps of homologous recombination. Mutations in the gene are associated with the disease Fanconi anemia.
Sister chromatid cohesion refers to the process by which sister chromatids are paired and held together during certain phases of the cell cycle. Establishment of sister chromatid cohesion is the process by which chromatin-associated cohesin protein becomes competent to physically bind together the sister chromatids. In general, cohesion is established during S phase as DNA is replicated, and is lost when chromosomes segregate during mitosis and meiosis. Some studies have suggested that cohesion aids in aligning the kinetochores during mitosis by forcing the kinetochores to face opposite cell poles.

Structural maintenance of chromosomes protein 1B (SMC-1B) is a protein that in humans is encoded by the SMC1B gene. SMC proteins engage in chromosome organization and can be broken into 3 groups based on function which are cohesins, condensins, and DNA repair.SMC-1B belongs to a family of proteins required for chromatid cohesion and DNA recombination during meiosis and mitosis. SMC1ß protein appears to participate with other cohesins REC8, STAG3 and SMC3 in sister-chromatid cohesion throughout the whole meiotic process in human oocytes.

Stromal antigen 3 is a protein that in humans is encoded by the STAG3 gene. STAG3 protein is a component of a cohesin complex that regulates the separation of sister chromatids specifically during meiosis. STAG3 appears to be paramount in sister-chromatid cohesion throughout the meiotic process in human oocytes and spermatocytes.
























