
Dipipanone, sold under the brand names of Pipadone and Diconal is a strong opioid analgesic drug, used for acute pain by mouth (PO) for adults. It is often used in instances where morphine is indicated but cannot be used due to the patient being allergic to morphine. In analgesic potency 25 mg dipipanone is approximately equivalent to 10 mg morphine.

Levomethorphan (LVM) (INN, BAN) is an opioid analgesic of the morphinan family that has never been marketed. It is the L-stereoisomer of racemethorphan (methorphan). The effects of the two isomers of racemethorphan are quite different, with dextromethorphan (DXM) being an antitussive at low doses and a dissociative hallucinogen at much higher doses. Levomethorphan is about five times stronger than morphine.

Piritramide(R-3365, trade names Dipidolor, Piridolan, Pirium and others) is a synthetic opioid analgesic that is marketed in certain European countries including: Austria, Belgium, Czech Republic, Slovenia, Germany and the Netherlands. It comes in free form, is about 0.75x times as potent as morphine and is given parenterally for the treatment of severe pain. Nausea, vomiting, respiratory depression and constipation are believed to be less frequent with piritramide than with morphine, and it produces more rapid-onset analgesia when compared to morphine and pethidine. After intravenous administration the onset of analgesia is as little as 1–2 minutes, which may be related to its great lipophilicity. The analgesic and sedative effects of piritramide are believed to be potentiated with phenothiazines and its emetic (nausea/vomiting-inducing) effects are suppressed. The volume of distribution is 0.7-1 L/kg after a single dose, 4.7-6 L/kg after steady-state concentrations are achieved and up to 11.1 L/kg after prolonged dosing.
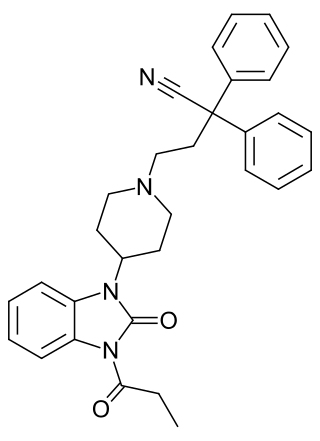
Bezitramide is an opioid analgesic. Bezitramide itself is a prodrug which is readily hydrolyzed in the gastrointestinal tract to its active metabolite, despropionyl-bezitramide. Bezitramide was discovered at Janssen Pharmaceutica in 1961. It is most commonly marketed under the trade name Burgodin.

Thebacon, or dihydrocodeinone enol acetate, is a semisynthetic opioid that is similar to hydrocodone and is most commonly synthesised from thebaine. Thebacon was invented in Germany in 1924, four years after the first synthesis of hydrocodone. Thebacon is a derivative of acetyldihydrocodeine, where only the 6–7 double bond is saturated. Thebacon is marketed as its hydrochloride salt under the trade name Acedicon, and as its bitartrate under Diacodin and other trade names. The hydrochloride salt has a free base conversion ratio of 0.846. Other salts used in research and other settings include thebacon's phosphate, hydrobromide, citrate, hydroiodide, and sulfate.

Nicocodeine is an opioid analgesic and cough suppressant, an ester of codeine closely related to dihydrocodeine and the codeine analogue of nicomorphine. It is not commonly used in most countries, but has activity similar to other opiates. Nicocodeine and nicomorphine were synthesized in 1904, and introduced in 1957 by Lannacher Heilmittel of Austria. Nicocodeine is metabolised in the liver by demethylation to produce nicomorphine, also known as 6-nicotinoylmorphine, and subsequently further metabolised to morphine. Side effects are similar to those of other opiates and include itching, nausea and respiratory depression. Related opioid analogues such as nicomorphine and nicodicodeine were first synthesized. The definitive synthesis, which involves treating anhydrous codeine base with nicotinic anhydride at 130 °C, was published by Pongratz and Zirm in Monatshefte für Chemie in 1957, simultaneously with the two analogues in an article about amides and esters of various organic acids.

Properidine is an opioid, an analgesic, and the isopropyl analog of pethidine. Properidine is under international control and is listed in the United States under the Controlled Substances Act (1970) as a Schedule I substance. It is a narcotic, with an Administrative Controlled Substances Code Number (ACSCN) of 9644 and a 2 gramme annual aggregate manufacturing quota as of 2014. The salt in use is hydrochloride, with a free base conversion ratio of 0.88.
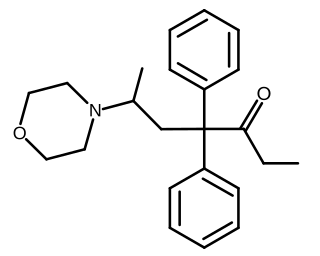
Phenadoxone is an opioid analgesic of the open chain class invented in Germany by Hoechst in 1947. It is one of a handful of useful synthetic analgesics which were used in the United States for various lengths of time in the 20 or so years after the end of the Second World War but which were withdrawn from the market for various or no known reason and which now are mostly in Schedule I of the United States' Controlled Substances Act of 1970, or in Schedule II but not produced or marketed in the US. Others on this list are ketobemidone (Ketogin), dextromoramide, phenazocine, dipipanone, piminodine (Alvodine), propiram (Algeril), anileridine (Leritine) and alphaprodine (Nisentil).

Benzylmorphine (Peronine) is a semi-synthetic opioid narcotic introduced to the international market in 1896 and that of the United States very shortly thereafter. It is much like codeine, containing a benzyl group attached to the morphine molecule just as the methyl group creates codeine and the ethyl group creates ethylmorphine or dionine. It is about 90% as strong as codeine by weight.

Ethylmethylthiambutene (N-ethyl-N-methyl-1-methyl-3,3-di-2-thienylallylamine; Emethibutin) is an opioid analgesic drug from the thiambutene family, around 1.3x the potency of morphine. It is under international control under Schedule I of the UN Single Convention On Narcotic Drugs 1961, presumably due to high abuse potential.

Normethadone, also known as desmethylmethadone or phenyldimazone, is a synthetic opioid analgesic and antitussive agent. Normethadone is listed under the Single Convention on Narcotic Drugs 1961 and is a Schedule I Narcotic controlled substance in the United States, with a DEA ACSCN of 9635 and an annual manufacturing quota of 2 grams. It has an effective span of action for about 14 days, and is 12 to 20 times stronger than morphine. The salts in use are the hydrobromide, hydrochloride (0.890), methyliodide (0.675), oxalate (0.766), picrate (0.563), and the 2,6-ditertbutylnapthalindisulphonate (0.480).
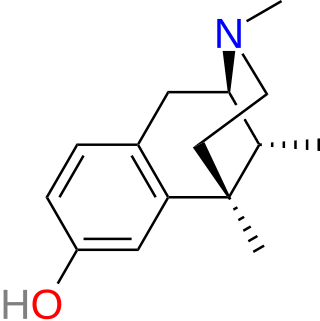
Metazocine is an opioid analgesic related to pentazocine. While metazocine has significant analgesic effects, mediated through a mixed agonist–antagonist action at the mu opioid receptor, its clinical use is limited by dysphoric and hallucinogenic effects which are most likely caused by activity at kappa opioid receptors and/or sigma receptors.

Phenazocine is an opioid analgesic drug, which is related to pentazocine and has a similar profile of effects.

Propiram is a partial μ-opioid receptor agonist and weak μ antagonist analgesic from the ampromide family of drugs related to other drugs such as phenampromide and diampromide. It was invented in 1963 in the United Kingdom by Bayer but was not widely marketed, although it saw some limited clinical use, especially in dentistry. Propiram reached Phase III clinical trials in the United States and Canada.

Phenampromide is an opioid analgesic from the ampromide family of drugs, related to other drugs such as propiram and diampromide. It was invented in the 1960s by American Cyanamid Co. Although never given a general release, it was research found that 60 mg of phenampromide is equivalent to about 50 mg of codeine. Tests on its two enantiomers showed that all of the analgesic effects were caused by the (S)-isomer. Introduction of a phenyl group to the 4-position of the piperidine-ring produces a drug 60-fold more potent than morphine. The most potent reported derivative is 4-hydroxy-4-phenyl phenapromide which displays analgesic activity some x150 greater than morphine.

Diampromide is an opioid analgesic from the ampromide family of drugs, related to other drugs such as propiram and phenampromide. It was invented in the 1960s by American Cyanamid, and can be described as a ring-opened analogue of fentanyl.
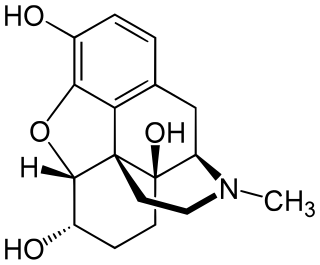
Hydromorphinol, is an opiate analogue that is a derivative of morphine, where the 14-position has been hydroxylated and the 7,8- double bond saturated. It has similar effects to morphine such as sedation, analgesia and respiratory depression, but is twice as potent as morphine and has a steeper dose-response curve and longer half-life. It is used in medicine as the bitartrate salt and hydrochloride

Normorphine is an opiate analogue, the N-demethylated derivative of morphine, that was first described in the 1950s when a large group of N-substituted morphine analogues were characterized for activity. The compound has relatively little opioid activity in its own right, but is a useful intermediate which can be used to produce both opioid antagonists such as nalorphine, and also potent opioid agonists such as N-phenethylnormorphine. with its formation from morphine catalyzed by the liver enzymes CYP3A4 and CYP2C8.

Methyldesorphine is an opioid analgesic. First synthesized in Germany in 1940 and patented in the US in 1952, it has a high potential for abuse as with any potent opioid agonist, and is sometimes found along with desomorphine as a component of the home-made opioid mixture known as "Krokodil" used in Russia and the neighboring former Soviet republics. It is approximately 15 times more potent than morphine as an analgesic but if the 6-7 bond is saturated, the β isomer is some 50 times more potent than morphine.

Noracymethadol (INN) is a synthetic opioid analgesic related to methadone that was never marketed. In a clinical trial of postpartum patients it was reported to produce analgesia comparable to that of morphine but with less nausea, dizziness, and drowsiness. Other side effects included salivation, ataxia, and respiratory depression that was reversible by naloxone. Similarly to many of its analogues, noracymethadol is a Schedule I controlled substance in the United States with an ACSCN of 9633 and 2013 annual manufacturing quota of 12 grammes. and is also controlled internationally under the United Nations Single Convention on Narcotic Drugs of 1961. The salts known are the gluconate and hydrochloride (0.903).




















