Coatomer subunit beta is a protein that in humans is encoded by the COPB1 gene. [5]
Coatomer subunit beta is a protein that in humans is encoded by the COPB1 gene. [5]

COPII is a coatomer, a type of vesicle coat protein that transports proteins from the rough endoplasmic reticulum to the Golgi apparatus. This process is termed anterograde transport, in contrast to the retrograde transport associated with the COPI protein. The name "COPII" refers to the specific coat protein complex that initiates the budding process. The coat consists of large protein subcomplexes that are made of four different protein subunits.

COPI is a coatomer, a protein complex that coats vesicles transporting proteins from the cis end of the Golgi complex back to the rough endoplasmic reticulum (ER), where they were originally synthesized, and between Golgi compartments. This type of transport is retrograde transport, in contrast to the anterograde transport associated with the COPII protein. The name "COPI" refers to the specific coat protein complex that initiates the budding process on the cis-Golgi membrane. The coat consists of large protein subcomplexes that are made of seven different protein subunits, namely α, β, β', γ, δ, ε and ζ.
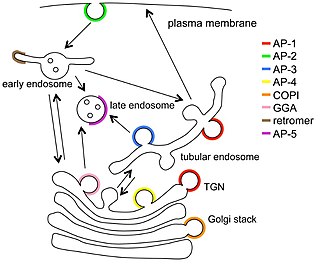
Vesicular transport adaptor proteins are proteins involved in forming complexes that function in the trafficking of molecules from one subcellular location to another. These complexes concentrate the correct cargo molecules in vesicles that bud or extrude off of one organelle and travel to another location, where the cargo is delivered. While some of the details of how these adaptor proteins achieve their trafficking specificity has been worked out, there is still much to be learned.
The coatomer is a protein complex that coats membrane-bound transport vesicles. Two types of coatomers are known:

ADP-ribosylation factor 1 is a protein that in humans is encoded by the ARF1 gene.

AP-2 complex subunit alpha-1 is a protein that in humans is encoded by the AP2A1 gene.

AP-1 complex subunit mu-1 is a protein that in humans is encoded by the AP1M1 gene.

Clathrin heavy chain 1 is a protein that in humans is encoded by the CLTC gene.

AP-1 complex subunit beta-1 is a protein that in humans is encoded by the AP1B1 gene.

Coatomer subunit alpha is a protein that in humans is encoded by the COPA gene.

Coatomer subunit epsilon is a protein that in humans is encoded by the COPE gene.

AP-1 complex subunit sigma-1A is a protein that in humans is encoded by the AP1S1 gene.

Coatomer subunit beta' is a protein that in humans is encoded by the COPB2 gene.

Coatomer subunit gamma-2 is a protein that in humans is encoded by the COPG2 gene.

Coatomer subunit gamma is a protein that in humans is encoded by the COPG gene. It is one of seven proteins in the COPI coatomer complex that coats vesicles as they bud from the Golgi complex.

AP-1 complex subunit gamma-like 2 is a protein that in humans is encoded by the AP1G2 gene.

AP-3 complex subunit mu-1 is a protein that in humans is encoded by the AP3M1 gene.

AP-1 complex subunit sigma-2 is a protein that in humans is encoded by the AP1S2 gene.
Clathrin adaptor proteins, also known as adaptins, are vesicular transport adaptor proteins associated with clathrin. These proteins are synthesized in the ribosomes, processed in the endoplasmic reticulum and transported from the Golgi apparatus to the trans-Golgi network, and from there via small carrier vesicles to their final destination compartment. The association between adaptins and clathrin are important for vesicular cargo selection and transporting. Clathrin coats contain both clathrin and adaptor complexes that link clathrin to receptors in coated vesicles. Clathrin-associated protein complexes are believed to interact with the cytoplasmic tails of membrane proteins, leading to their selection and concentration. Therefore, adaptor proteins are responsible for the recruitment of cargo molecules into a growing clathrin-coated pits. The two major types of clathrin adaptor complexes are the heterotetrameric vesicular transport adaptor proteins (AP1-5), and the monomeric GGA adaptors. Adaptins are distantly related to the other main type of vesicular transport proteins, the coatomer subunits, sharing between 16% and 26% of their amino acid sequence.

The C-terminal domain ofBeta2-adaptin is a protein domain is involved in cell trafficking by aiding import and export of substances in and out of the cell.