Vesicle-associated membrane protein 2 (VAMP2) is a protein that in humans is encoded by the VAMP2 gene. [5] [6]

Vesicle-associated membrane protein 2 (VAMP2) is a protein that in humans is encoded by the VAMP2 gene. [5] [6]
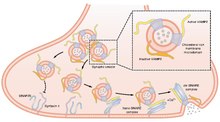
Synaptobrevins/VAMPs, syntaxins, and the 25-kD synaptosomal-associated protein SNAP25 are the main components of a protein complex involved in the docking and/or fusion of synaptic vesicles with the presynaptic membrane. VAMP2 is a member of the vesicle-associated membrane protein (VAMP)/synaptobrevin family. VAMP2 is thought to participate in neurotransmitter release at a step between docking and fusion. Mice lacking functional synaptobrevin2/VAMP2 gene cannot survive after birth, and have a dramatically reduced synaptic transmission, around 10% of control. [7] The protein forms a stable complex with syntaxin, synaptosomal-associated protein, 25 kD, and complexin. It also forms a distinct complex with synaptophysin. [6]
Heterozygous mutations in VAMP2 cause a neurodevelopmental disorder with hypotonia and autistic features (with or without hyperkinetic movements). [8] [9] [10]
VAMP2 has been shown to interact with:

SNARE proteins – "SNAPREceptors" – are a large protein family consisting of at least 24 members in yeasts, more than 60 members in mammalian cells, and some numbers in plants. The primary role of SNARE proteins is to mediate the fusion of vesicles with the target membrane; this notably mediates exocytosis, but can also mediate the fusion of vesicles with membrane-bound compartments. The best studied SNAREs are those that mediate the release of synaptic vesicles containing neurotransmitters in neurons. These neuronal SNAREs are the targets of the neurotoxins responsible for botulism and tetanus produced by certain bacteria.
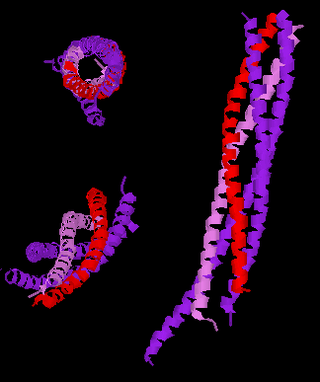
Synaptobrevins are small integral membrane proteins of secretory vesicles with molecular weight of 18 kilodalton (kDa) that are part of the vesicle-associated membrane protein (VAMP) family.

Synaptosomal-Associated Protein, 25kDa (SNAP-25) is a Target Soluble NSF (N-ethylmaleimide-sensitive factor) Attachment Protein Receptor (t-SNARE) protein encoded by the SNAP25 gene found on chromosome 20p12.2 in humans. SNAP-25 is a component of the trans-SNARE complex, which accounts for membrane fusion specificity and directly executes fusion by forming a tight complex that brings the synaptic vesicle and plasma membranes together.

Syntaxin-1A is a protein that in humans is encoded by the STX1A gene.

Synaptosomal-associated protein 23 is a protein that in humans is encoded by the SNAP23 gene. Two alternative transcript variants encoding different protein isoforms have been described for this gene.

Syntaxin-4 is a protein that in humans is encoded by the STX4 gene.

Synaptotagmin-1 is a protein that in humans is encoded by the SYT1 gene.

Syntaxin-binding protein 1 is a protein that in humans is encoded by the STXBP1 gene. This gene encodes a syntaxin-binding protein. The encoded protein appears to play a role in release of neurotransmitters via regulation of syntaxin, a transmembrane attachment protein receptor. Mutations in this gene have been associated with neurological disorders including epilepsy, intellectual disability, and movement disorders.

Vesicle-associated membrane protein 7 (VAMP-7), is a protein that in humans is encoded by the VAMP7 gene also known as the or SYBL1 gene.

Syntaxin-7 is a protein that in humans is encoded by the STX7 gene.

N-ethylmaleimide-sensitive factor Attachment Protein Alpha, also known as SNAP-α, is a SNAP protein that is involved in the intra-cellular trafficking and fusing of vesicles to target membranes in cells.

Vesicle-associated membrane protein 3 is a protein that in humans is encoded by the VAMP3 gene.

Syntaxin-2, also known as epimorphin, is a protein that in humans is encoded by the STX2 gene.

Vesicle-associated membrane protein 8 is a protein that in humans is encoded by the VAMP8 gene.

Vesicle-associated membrane protein 4 is a protein that in humans is encoded by the VAMP4 gene.

Vesicle-associated membrane protein 1 (VAMP1) is a protein that in humans is encoded by the VAMP1 gene.

Syntaxin 3, also known as STX3, is a protein which in humans is encoded by the STX3 gene.

Syntaxins are a family of membrane integrated Q-SNARE proteins participating in exocytosis.
Munc-18 proteins are the mammalian homologue of UNC-18 and are a member of the Sec1/Munc18-like (SM) protein family. Munc-18 proteins have been identified as essential components of the synaptic vesicle fusion protein complex and are crucial for the regulated exocytosis of neurons and neuroendocrine cells.

Soluble N-ethylmaleimide-Sensitive Factor Attachment Proteins are a family of cytosolic adaptor proteins involved in vesicular fusion at membranes during intracellular transport and exocytosis. SNAPs interact with proteins of the SNARE complex and NSF to play a key role in recycling the components of the fusion complex. SNAPs are involved in the priming of the vesicle fusion complex during assembly, as well as in the disassembly following a vesicle fusion event. Following membrane fusion, the tethering SNARE proteins complex disassembles in response to steric changes originating from the ATPase NSF. The energy provided by NSF is transferred throughout the SNARE complex and SNAP, allowing the proteins to untangle, and recycled for future fusion events. Mammals have three SNAP genes: α-SNAP, β-SNAP, and γ-SNAP. α- and γ-SNAP are expressed throughout the body, while β-SNAP is specific to the brain. The yeast homolog of the human SNAP is Sec17, the structural diagram of which is included on this page.