
Dichlorodiphenyltrichloroethane, commonly known as DDT, is a colorless, tasteless, and almost odorless crystalline chemical compound, an organochloride. Originally developed as an insecticide, it became infamous for its environmental impacts. DDT was first synthesized in 1874 by the Austrian chemist Othmar Zeidler. DDT's insecticidal action was discovered by the Swiss chemist Paul Hermann Müller in 1939. DDT was used in the second half of World War II to limit the spread of the insect-borne diseases malaria and typhus among civilians and troops. Müller was awarded the Nobel Prize in Physiology or Medicine in 1948 "for his discovery of the high efficiency of DDT as a contact poison against several arthropods". The WHO's anti-malaria campaign of the 1950s and 1960s relied heavily on DDT and the results were promising, though there was a resurgence in developing countries afterwards.

Insecticides are pesticides used to kill insects. They include ovicides and larvicides used against insect eggs and larvae, respectively. Acaricides, which kill mites and ticks, are not strictly insecticides, but are usually classified together with insecticides. The major use of Insecticides is agriculture, but they are also used in home and garden, industrial buildings, vector control and control of insect parasites of animals and humans. Insecticides are claimed to be a major factor behind the increase in the 20th-century's agricultural productivity. Nearly all insecticides have the potential to significantly alter ecosystems; many are toxic to humans and/or animals; some become concentrated as they spread along the food chain.
Pyrethrum was a genus of several Old World plants now classified in either Chrysanthemum or Tanacetum which are cultivated as ornamentals for their showy flower heads. Pyrethrum continues to be used as a common name for plants formerly included in the genus Pyrethrum. Pyrethrum is also the name of a natural insecticide made from the dried flower heads of Chrysanthemum cinerariifolium and Chrysanthemum coccineum. The insecticidal compounds present in these species are pyrethrins.

The pyrethrins are a class of organic compounds normally derived from Chrysanthemum cinerariifolium that have potent insecticidal activity by targeting the nervous systems of insects. Pyrethrin naturally occurs in chrysanthemum flowers and is often considered an organic insecticide when it is not combined with piperonyl butoxide or other synthetic adjuvants. Their insecticidal and insect-repellent properties have been known and used for thousands of years.

Fenobucarb is a carbamate insecticide, also widely known as BPMC. A pale yellow or pale red liquid, insoluble in water; used as an agricultural insecticide, especially for control of Hemipteran pests, on rice and cotton and moderately toxic for humans.

Fenvalerate is a synthetic pyrethroid insecticide. It is a mixture of four optical isomers which have different insecticidal activities. The 2-S alpha configuration, known as esfenvalerate, is the most insecticidally active isomer. Fenvalerate consists of about 23% of this isomer.

Carbaryl is a chemical in the carbamate family used chiefly as an insecticide. It is a white crystalline solid previously sold under the brand name Sevin, which was a trademark of the Bayer Company. The Sevin trademark has since been acquired by GardenTech, which has eliminated carbaryl from most Sevin formulations. Union Carbide discovered carbaryl and introduced it commercially in 1958. Bayer purchased Aventis CropScience in 2002, a company that included Union Carbide pesticide operations. Carbaryl was the third-most-used insecticide in the United States for home gardens, commercial agriculture, and forestry and rangeland protection. As a veterinary drug, it is known as carbaril (INN).
Hydantoin, or glycolylurea, is a heterocyclic organic compound with the formula CH2C(O)NHC(O)NH. It is a colorless solid that arises from the reaction of glycolic acid and urea. It is an oxidized derivative of imidazolidine. In a more general sense, hydantoins can refer to groups or a class of compounds with the same ring structure as the parent compound. For example, phenytoin (mentioned below) has two phenyl groups substituted onto the number 5 carbon in a hydantoin molecule.

Isoxathion is a molecular chemical with the molecular formula C13H16NO4PS. It is an insecticide, specifically an isoxazole organothiophosphate insecticide.

A mosquito net is a type of meshed curtain that is circumferentially draped over a bed or a sleeping area, to offer the sleeper barrier protection against bites and stings from mosquitos, flies, and other pest insects, and thus against the diseases they may carry. Examples of such preventable insect-borne diseases include malaria, dengue fever, yellow fever, zika virus, Chagas disease and various forms of encephalitis, including the West Nile virus.

Tetramethrin is a potent synthetic insecticide in the pyrethroid family. It is a white crystalline solid with a melting point of 65–80 °C. The commercial product is a mixture of stereoisomers.

Deltamethrin is a pyrethroid ester insecticide. Deltamethrin plays a key role in controlling malaria vectors, and is used in the manufacture of long-lasting insecticidal mosquito nets; however, resistance of mosquitos and bed bugs to deltamethrin has seen a widespread increase.

Spinosad is an insecticide based on chemical compounds found in the bacterial species Saccharopolyspora spinosa. The genus Saccharopolyspora was discovered in 1985 in isolates from crushed sugarcane. The bacteria produce yellowish-pink aerial hyphae, with bead-like chains of spores enclosed in a characteristic hairy sheath. This genus is defined as aerobic, Gram-positive, nonacid-fast actinomycetes with fragmenting substrate mycelium. S. spinosa was isolated from soil collected inside a nonoperational sugar mill rum still in the Virgin Islands. Spinosad is a mixture of chemical compounds in the spinosyn family that has a generalized structure consisting of a unique tetracyclic ring system attached to an amino sugar (D-forosamine) and a neutral sugar (tri-Ο-methyl-L-rhamnose). Spinosad is relatively nonpolar and not easily dissolved in water.

Paraoxon is a parasympathomimetic which acts as an cholinesterase inhibitor. It is an organophosphate oxon, and the active metabolite of the insecticide parathion. It is also used as an ophthalmological drug against glaucoma. Paraoxon is one of the most potent acetylcholinesterase-inhibiting insecticides available, around 70% as potent as the nerve agent sarin, and so is now rarely used as an insecticide due to the risk of poisoning to humans and other animals. Paraoxon has been used by scientists to study acute and chronic effects of organophosphate intoxication. It is easily absorbed through skin, and was allegedly used as an assassination weapon by the apartheid-era South African chemical weapons program Project Coast.
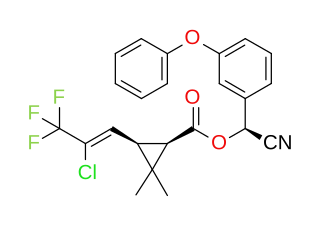
Cyhalothrin is the ISO common name for an organic compound that, in specific isomeric forms, is used as a pesticide. It is a pyrethroid, a class of synthetic insecticides that mimic the structure and properties of the naturally occurring insecticide pyrethrin which is present in the flowers of Chrysanthemum cinerariifolium. Pyrethroids such as cyhalothrin are often preferred as an active ingredient in agricultural insecticides because they are more cost-effective and longer acting than natural pyrethrins. λ-and γ-cyhalothrin are now used to control insects and spider mites in crops including cotton, cereals, potatoes and vegetables.

Indoxacarb is an oxadiazine pesticide developed by DuPont that acts against lepidopteran larvae. It is marketed under the names Indoxacarb Technical Insecticide, Steward Insecticide and Avaunt Insecticide. It is also used as the active ingredient in the Syngenta line of commercial pesticides: Advion and Arilon.

Cyphenothrin is a synthetic pyrethroid insecticide. It is effective against cockroaches that have developed resistance to organophosphorous and carbamate insecticides.
Ryanoids are a class of insecticides which share the same mechanism of action as the alkaloid ryanodine. Ryanodine is a naturally occurring insecticide isolated from Ryania speciosa.

Cyantraniliprole is an insecticide of the ryanoid class, specifically a diamide insecticide. It is approved for use in the United States, Canada, China, and India. Because of its uncommon mechanism of action as a ryanoid, it has activity against pests such as Diaphorina citri that have developed resistance to other classes of insecticides.

Difluorodiphenyltrichloroethane (DFDT) is a chemical compound. Its composition is the same as that of the insecticide DDT, except that two of DDT's chlorine atoms are replaced by two fluorine atoms.



















