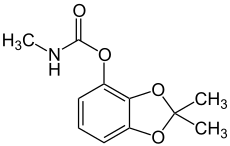
Pesticides are substances that are meant to control pests. This includes herbicide, insecticide, nematicide, molluscicide, piscicide, avicide, rodenticide, bactericide, insect repellent, animal repellent, microbicide, fungicide, and lampricide. The most common of these are herbicides, which account for approximately 50% of all pesticide use globally. Most pesticides are intended to serve as plant protection products, which in general, protect plants from weeds, fungi, or insects. In general, a pesticide is a chemical or biological agent that deters, incapacitates, kills, or otherwise discourages pests. Target pests can include insects, plant pathogens, weeds, molluscs, birds, mammals, fish, nematodes (roundworms), and microbes that destroy property, cause nuisance, or spread disease, or are disease vectors. Along with these benefits, pesticides also have drawbacks, such as potential toxicity to humans and other species.

Insecticides are pesticides used to kill insects. They include ovicides and larvicides used against insect eggs and larvae, respectively. Insecticides are used in agriculture, medicine, industry and by consumers. Insecticides are claimed to be a major factor behind the increase in the 20th-century's agricultural productivity. Nearly all insecticides have the potential to significantly alter ecosystems; many are toxic to humans and/or animals; some become concentrated as they spread along the food chain.

Cypermethrin (CP) is a synthetic pyrethroid used as an insecticide in large-scale commercial agricultural applications as well as in consumer products for domestic purposes. It behaves as a fast-acting neurotoxin in insects. It is easily degraded on soil and plants but can be effective for weeks when applied to indoor inert surfaces. Exposure to sunlight, water and oxygen will accelerate its decomposition. Cypermethrin is highly toxic to fish, bees and aquatic insects, according to the National Pesticides Telecommunications Network (NPTN). It is found in many household ant and cockroach killers, including Raid, Ortho, Combat, ant chalk, and some products of Baygon in Southeast Asia.

Piperonyl butoxide (PBO) is a pale yellow to light brown liquid organic compound used as a synergist component of pesticide formulations. That is, despite having no pesticidal activity of its own, it enhances the potency of certain pesticides such as carbamates, pyrethrins, pyrethroids, and rotenone. It is a semisynthetic derivative of safrole.

The pyrethrins are a class of organic compounds normally derived from Chrysanthemum cinerariifolium that have potent insecticidal activity by targeting the nervous systems of insects. Pyrethrin naturally occurs in chrysanthemum flowers and is often considered an organic insecticide when it is not combined with piperonyl butoxide or other synthetic adjuvants. Their insecticidal and insect-repellent properties have been known and used for thousands of years.

Bifenthrin is a pyrethroid insecticide. It is widely used against ant infestations.

Carbofuran is a carbamate pesticide, widely used around the world to control insects on a wide variety of field crops, including potatoes, corn and soybeans. It is a systemic insecticide, which means that the plant absorbs it through the roots, and from there the plant distributes it throughout its organs where insecticidal concentrations are attained. Carbofuran also has contact activity against pests. It is one of the most toxic pesticides still in use.

Omethoate (C5H12NO4PS) is a systemic organophosphorous insecticide and acaricide available as a soluble concentrate. It is used to control insects and mites in horticulture and agriculture, as well as in the home garden.

Carbaryl is a chemical in the carbamate family used chiefly as an insecticide. It is a white crystalline solid previously sold under the brand name Sevin, which was a trademark of the Bayer Company. The Sevin trademark has since been acquired by GardenTech, which has eliminated carbaryl from most Sevin formulations. Union Carbide discovered carbaryl and introduced it commercially in 1958. Bayer purchased Aventis CropScience in 2002, a company that included Union Carbide pesticide operations. Carbaryl was the third-most-used insecticide in the United States for home gardens, commercial agriculture, and forestry and rangeland protection. As a veterinary drug, it is known as carbaril (INN).

Imidacloprid is a systemic insecticide belonging to a class of chemicals called the neonicotinoids which act on the central nervous system of insects. The chemical works by interfering with the transmission of stimuli in the insect nervous system. Specifically, it causes a blockage of the nicotinergic neuronal pathway. By blocking nicotinic acetylcholine receptors, imidacloprid prevents acetylcholine from transmitting impulses between nerves, resulting in the insect's paralysis and eventual death. It is effective on contact and via stomach action. Because imidacloprid binds much more strongly to insect neuron receptors than to mammal neuron receptors, this insecticide is more toxic to insects than to mammals.

Chlorfenvinphos is an organophosphorus compound that was widely used as an insecticide and an acaricide. The molecule itself can be described as an enol ester derived from dichloroacetophenone and diethylphosphonic acid. Chlorfenvinphos has been included in many products since its first use in 1963. However, because of its toxic effect as a cholinesterase inhibitor it has been banned in several countries, including the United States and the European Union. Its use in the United States was cancelled in 1991.

Diazinon, a colorless to dark brown liquid, is a thiophosphoric acid ester developed in 1952 by Ciba-Geigy, a Swiss chemical company. It is a nonsystemic organophosphate insecticide formerly used to control cockroaches, silverfish, ants, and fleas in residential, non-food buildings. Diazinon was heavily used during the 1970s and early 1980s for general-purpose gardening use and indoor pest control. A bait form was used to control scavenger wasps in the western U.S. Diazinon is used in flea collars for domestic pets in Australia and New Zealand. Residential uses of diazinon were outlawed in the U.S. in 2004 because of human health risks but it is still approved for agricultural uses. An emergency antidote is atropine.
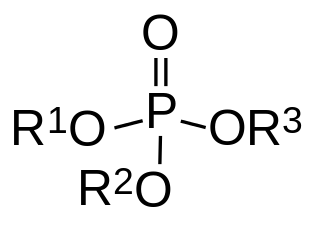
Organophosphate poisoning is poisoning due to organophosphates (OPs). Organophosphates are used as insecticides, medications, and nerve agents. Symptoms include increased saliva and tear production, diarrhea, vomiting, small pupils, sweating, muscle tremors, and confusion. While onset of symptoms is often within minutes to hours, some symptoms can take weeks to appear. Symptoms can last for days to weeks.

Methomyl is a carbamate insecticide introduced in 1966. It is highly toxic to humans, livestock, pets, and wildlife. The EU and UK imposed a pesticide residue limit of 20 µg/kg for apples and oranges.

Clothianidin is an insecticide developed by Takeda Chemical Industries and Bayer AG. Similar to thiamethoxam and imidacloprid, it is a neonicotinoid. Neonicotinoids are a class of insecticides that are chemically similar to nicotine, which has been used as a pesticide since the late 1700s. Clothianidin and other neonicotinoids act on the central nervous system of insects as an agonist of nAChR, the same receptor as acetylcholine, the neurotransmitter that stimulates and activating post-synaptic acetylcholine receptors but not inhibiting AChE. Clothianidin and other neonicotinoids were developed to last longer than nicotine, which is more toxic and which breaks down too quickly in the environment.

Disulfoton is an organophosphate acetylcholinesterase inhibitor used as an insecticide. It is manufactured under the name Di-Syston by Bayer CropScience. Disulfoton in its pure form is a colorless oil but the technical product used in vegetable fields is dark and yellowish with a sulfur odor. Disulfoton is processed as a liquid into carrier granules, these granules are mixed with fertilizer and clay to be made into a spike, designed to be driven into the ground. The pesticide is absorbed over time by the roots and translocated to all parts of the plant. The pesticide acts as a cholinesterase inhibitor and gives long lasting control.

Propoxur (Baygon) is a carbamate non-systemic insecticide, produced from catechol, and was introduced in 1959. It has a fast knockdown and long residual effect, and is used against turf, forestry, and household pests and fleas. It is also used in pest control for domestic animals, Anopheles mosquitoes, ants, gypsy moths, and other agricultural pests. It can also be used as a molluscicide.

Acetamiprid is an organic compound with the chemical formula C10H11ClN4. It is an odorless neonicotinoid insecticide produced under the trade names Assail, and Chipco by Aventis CropSciences. It is systemic and intended to control sucking insects (Thysanoptera, Hemiptera, mainly aphids) on crops such as leafy vegetables, citrus fruits, pome fruits, grapes, cotton, cole crops, and ornamental plants. It is also a key pesticide in commercial cherry farming due to its effectiveness against the larvae of the cherry fruit fly.

Terbufos is a chemical compound used in insecticides and nematicides. It is part of the chemical family of organophosphates. It is a clear, colourless to pale yellow or reddish-brown liquid and sold commercially as granulate.

Sulfoxaflor, also marketed as Isoclast, is a systemic insecticide that acts as an insect neurotoxin. A pyridine and a trifluoromethyl compound, it is a member of a class of chemicals called sulfoximines, which act on the central nervous system of insects.
Anisha, N.S.; Tumul, S., (2019). Evaluation of Genotoxic and Cytotoxic effects of BendioCarb in Bone Marrow cells of one Calotes Versicolor. Proceedings of the Zoological Society of India 18(1): 19-23