
Acetylcholine (ACh) is an organic compound that functions in the brain and body of many types of animals as a neurotransmitter. Its name is derived from its chemical structure: it is an ester of acetic acid and choline. Parts in the body that use or are affected by acetylcholine are referred to as cholinergic.
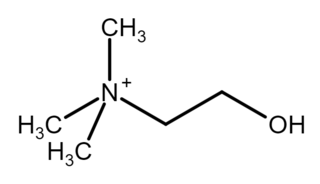
Choline is a cation with the chemical formula [(CH3)3NCH2CH2OH]+. Choline forms various salts, for example choline chloride and choline bitartrate.

Monoamine transporters (MATs) are proteins that function as integral plasma-membrane transporters to regulate concentrations of extracellular monoamine neurotransmitters. The three major classes are serotonin transporters (SERTs), dopamine transporters (DATs), and norepinephrine transporters (NETs) and are responsible for the reuptake of their associated amine neurotransmitters. MATs are located just outside the synaptic cleft (peri-synaptically), transporting monoamine transmitter overflow from the synaptic cleft back to the cytoplasm of the pre-synaptic neuron. MAT regulation generally occurs through protein phosphorylation and post-translational modification. Due to their significance in neuronal signaling, MATs are commonly associated with drugs used to treat mental disorders as well as recreational drugs. Compounds targeting MATs range from medications such as the wide variety of tricyclic antidepressants, selective serotonin reuptake inhibitors such as fluoxetine (Prozac) to stimulant medications such as methylphenidate (Ritalin) and amphetamine in its many forms and derivatives methamphetamine (Desoxyn) and lisdexamfetamine (Vyvanse). Furthermore, drugs such as MDMA and natural alkaloids such as cocaine exert their effects in part by their interaction with MATs, by blocking the transporters from mopping up dopamine, serotonin, and other neurotransmitters from the synapse.

Choline acetyltransferase is a transferase enzyme responsible for the synthesis of the neurotransmitter acetylcholine. ChAT catalyzes the transfer of an acetyl group from the coenzyme acetyl-CoA to choline, yielding acetylcholine (ACh). ChAT is found in high concentration in cholinergic neurons, both in the central nervous system (CNS) and peripheral nervous system (PNS). As with most nerve terminal proteins, ChAT is produced in the body of the neuron and is transported to the nerve terminal, where its concentration is highest. Presence of ChAT in a nerve cell classifies this cell as a "cholinergic" neuron. In humans, the choline acetyltransferase enzyme is encoded by the CHAT gene.

In a neuron, synaptic vesicles store various neurotransmitters that are released at the synapse. The release is regulated by a voltage-dependent calcium channel. Vesicles are essential for propagating nerve impulses between neurons and are constantly recreated by the cell. The area in the axon that holds groups of vesicles is an axon terminal or "terminal bouton". Up to 130 vesicles can be released per bouton over a ten-minute period of stimulation at 0.2 Hz. In the visual cortex of the human brain, synaptic vesicles have an average diameter of 39.5 nanometers (nm) with a standard deviation of 5.1 nm.
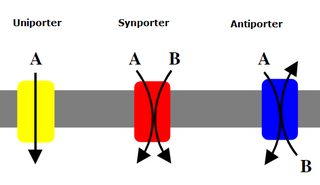
Uniporters, also known as solute carriers or facilitated transporters, are a type of membrane transport protein that passively transport solutes across a cell membrane. It uses facilitated diffusion for the movement of solutes down their concentration gradient, from an area of high concentration to an area of low concentration. Unlike active transport, it does not require energy in the form of ATP to function. Uniporters are specialized to carry one specific ion or molecule and can be categorized as either channels or carriers. Facilitated diffusion may occur through three mechanisms: uniport, symport, or antiport. The difference between each mechanism depends on the direction of transport, in which uniport is the only transport not coupled to the transport of another solute.

Coluracetam is a purported nootropic agent of the racetam family. It is contains a chemical group that is a bioisostere of the 9-amino-tetrahydroacridine family. It was initially developed and tested by the Mitsubishi Tanabe Pharma Corporation for Alzheimer's disease. After the drug failed to reach endpoints in its clinical trials it was in-licensed by BrainCells Inc for investigations into major depressive disorder (MDD), which was preceded by being awarded a "Qualifying Therapeutic Discovery Program Grant" by the state of California. Findings from phase IIa clinical trials have suggested that it would be a potential medication for comorbid MDD with generalized anxiety disorder (GAD). BrainCells Inc is currently out-licensing the drug for this purpose. It may also have potential use in prevention and treatment of ischemic retinopathy and retinal and optic nerve injury.
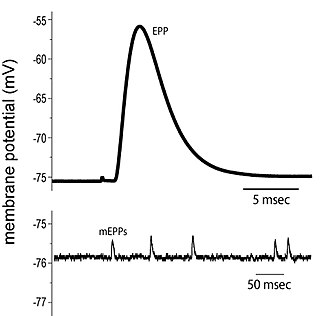
End plate potentials (EPPs) are the voltages which cause depolarization of skeletal muscle fibers caused by neurotransmitters binding to the postsynaptic membrane in the neuromuscular junction. They are called "end plates" because the postsynaptic terminals of muscle fibers have a large, saucer-like appearance. When an action potential reaches the axon terminal of a motor neuron, vesicles carrying neurotransmitters are exocytosed and the contents are released into the neuromuscular junction. These neurotransmitters bind to receptors on the postsynaptic membrane and lead to its depolarization. In the absence of an action potential, acetylcholine vesicles spontaneously leak into the neuromuscular junction and cause very small depolarizations in the postsynaptic membrane. This small response (~0.4mV) is called a miniature end plate potential (MEPP) and is generated by one acetylcholine-containing vesicle. It represents the smallest possible depolarization which can be induced in a muscle.

The dopamine transporter (DAT) also is a membrane-spanning protein coded for in the human by the SLC6A3 gene,, that pumps the neurotransmitter dopamine out of the synaptic cleft back into cytosol. In the cytosol, other transporters sequester the dopamine into vesicles for storage and later release. Dopamine reuptake via DAT provides the primary mechanism through which dopamine is cleared from synapses, although there may be an exception in the prefrontal cortex, where evidence points to a possibly larger role of the norepinephrine transporter.

The norepinephrine transporter (NET), also known as noradrenaline transporter (NAT), is a protein that in humans is encoded by the solute carrier family 6 member 2 (SLC6A2) gene.
An autoreceptor is a type of receptor located in the membranes of nerve cells. It serves as part of a negative feedback loop in signal transduction. It is only sensitive to the neurotransmitters or hormones released by the neuron on which the autoreceptor sits. Similarly, a heteroreceptor is sensitive to neurotransmitters and hormones that are not released by the cell on which it sits. A given receptor can act as either an autoreceptor or a heteroreceptor, depending upon the type of transmitter released by the cell on which it is embedded.
Glutamate transporters are a family of neurotransmitter transporter proteins that move glutamate – the principal excitatory neurotransmitter – across a membrane. The family of glutamate transporters is composed of two primary subclasses: the excitatory amino acid transporter (EAAT) family and vesicular glutamate transporter (VGLUT) family. In the brain, EAATs remove glutamate from the synaptic cleft and extrasynaptic sites via glutamate reuptake into glial cells and neurons, while VGLUTs move glutamate from the cell cytoplasm into synaptic vesicles. Glutamate transporters also transport aspartate and are present in virtually all peripheral tissues, including the heart, liver, testes, and bone. They exhibit stereoselectivity for L-glutamate but transport both L-aspartate and D-aspartate.

Hemicholinium-3 (HC3), also known as hemicholine, is a drug which blocks the reuptake of choline by the high-affinity choline transporter at the presynapse. The reuptake of choline is the rate-limiting step in the synthesis of acetylcholine; hence, hemicholinium-3 decreases the synthesis of acetylcholine. It is therefore classified as an indirect acetylcholine antagonist.

The Vesicular acetylcholine transporter (VAChT) is a neurotransmitter transporter which is responsible for loading acetylcholine (ACh) into secretory organelles in neurons making acetylcholine available for secretion. It is encoded by Solute carrier family 18, member 3 (SLC18A3) gene, located within the first intron of the choline acetyltransferase gene. VAChT is able to transport ACh into vesicles by relying on an exchange between protons (H+) that were previously pumped into the vesicle diffusing out, thus acting as an antiporter. ACh molecules are then carried into the vesicle by the action of exiting protons. Acetylcholine transport utilizes a proton gradient established by a vacuolar ATPase.

Natural resistance-associated macrophage protein 2, also known as divalent metal transporter 1 (DMT1) and divalent cation transporter 1 (DCT1), is a protein that in humans is encoded by the SLC11A2 gene. DMT1 represents a large family of orthologous metal ion transporter proteins that are highly conserved from bacteria to humans.

GABA transporter 1 (GAT1) also known as sodium- and chloride-dependent GABA transporter 1 is a protein that in humans is encoded by the SLC6A1 gene and belongs to the solute carrier 6 (SLC6) family of transporters. It mediates gamma-aminobutyric acid's translocation from the extracellular to intracellular spaces within brain tissue and the central nervous system as a whole.
Neuromuscular junction disease is a medical condition where the normal conduction through the neuromuscular junction fails to function correctly.

The plasma membrane monoamine transporter (PMAT) is a low-affinity monoamine transporter protein which in humans is encoded by the SLC29A4 gene. It is known alternatively as the human equilibrative nucleoside transporter-4 (hENT4). Unlike other members of the ENT family, it is impermeable to most nucleosides, with the exception of the inhibitory neurotransmitter and ribonucleoside adenosine, which it is permeable to in a highly pH-dependent manner.

The CDP-choline pathway, first identified by Eugene P. Kennedy in 1956, is the predominant mechanism by which mammalian cells synthesize phosphatidylcholine (PC) for incorporation into membranes or lipid-derived signalling molecules. The CDP-choline pathway represents one half of what is known as the Kennedy pathway. The other half is the CDP-ethanolamine pathway which is responsible for the biosynthesis of the phospholipid phosphatidylethanolamine (PE).
Notexin is a toxin produced by the tiger snake (Notechis scutatus). It is a myotoxic and presynaptic, neurotoxic phospholipase A2 (PLA2s). These are enzymes that hydrolyze the bond between a fatty acid tail and glycerol in fatty acids on the 2-position.



















