- Part of the polymeric structure of LiPPh2(Et2O). [5]
 | |
| Names | |
|---|---|
| Preferred IUPAC name Lithium diphenylphosphanide | |
| Identifiers | |
3D model (JSmol) | |
| ChemSpider | |
PubChem CID | |
CompTox Dashboard (EPA) | |
| |
| |
| Properties | |
| C12H10LiP | |
| Molar mass | 192.13 g·mol−1 |
| Appearance | pale yellow solid |
| Reacts with water | |
| Solubility | Ethers, hydrocarbons |
| Hazards | |
| GHS labelling: | |
   | |
| Danger | |
| H302, H312, H314, H332, H410 | |
| P260, P261, P264, P270, P271, P273, P280, P301+P312, P301+P330+P331, P302+P352, P303+P361+P353, P304+P312, P304+P340, P305+P351+P338, P310, P312, P321, P322, P330, P363, P391, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
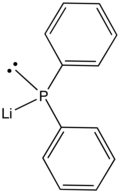
Lithium diphenylphosphide contains lithium and the organophosphorus anion with the formula (C6H5)2PLi. It is a red, air-sensitive solid that is used in the preparation of diphenylphosphino compounds.
