A catecholamine is a monoamine neurotransmitter, an organic compound that has a catechol and a side-chain amine.

Catechol-O-methyltransferase is one of several enzymes that degrade catecholamines, catecholestrogens, and various drugs and substances having a catechol structure. In humans, catechol-O-methyltransferase protein is encoded by the COMT gene. Two isoforms of COMT are produced: the soluble short form (S-COMT) and the membrane bound long form (MB-COMT). As the regulation of catecholamines is impaired in a number of medical conditions, several pharmaceutical drugs target COMT to alter its activity and therefore the availability of catecholamines. COMT was first discovered by the biochemist Julius Axelrod in 1957.

5-Hydroxytryptophan (5-HTP), also known as oxitriptan, is a naturally occurring amino acid and chemical precursor as well as a metabolic intermediate in the biosynthesis of the neurotransmitter serotonin.
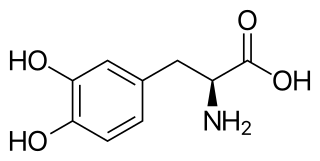
l-DOPA, also known as levodopa and l-3,4-dihydroxyphenylalanine, is made and used as part of the normal biology of some plants and animals, including humans. Humans, as well as a portion of the other animals that utilize l-DOPA, make it via biosynthesis from the amino acid l-tyrosine. l-DOPA is the precursor to the neurotransmitters dopamine, norepinephrine (noradrenaline), and epinephrine (adrenaline), which are collectively known as catecholamines. Furthermore, l-DOPA itself mediates neurotrophic factor release by the brain and CNS. In some plant families, l-DOPA is the central precursor of a biosynthetic pathway that produces a class of pigments called betalains. l-DOPA can be manufactured and in its pure form is sold as a psychoactive drug with the INN levodopa; trade names include Sinemet, Pharmacopa, Atamet, and Stalevo. As a drug, it is used in the clinical treatment of Parkinson's disease and dopamine-responsive dystonia.
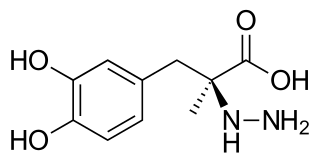
Carbidopa (Lodosyn) is a drug given to people with Parkinson's disease in order to inhibit peripheral metabolism of levodopa. This property is significant in that it allows a greater proportion of administered levodopa to cross the blood–brain barrier for central nervous system effect, instead of being peripherally metabolised into substances unable to cross said barrier.

Zafirlukast is an orally administered leukotriene receptor antagonist (LTRA) used for the chronic treatment of asthma. While zafirlukast is generally well tolerated, headache and stomach upset often occur. Some rare side effects can occur, which can be life-threatening, such as liver failure. Churg-Strauss syndrome has been associated with zafirlukast, but the relationship isn't thought to be causative in nature. Overdoses of zafirlukast tend to be self-limiting.
Carbidopa/levodopa, also known as levocarb and co-careldopa, is the combination of the two medications carbidopa and levodopa. It is primarily used to manage the symptoms of Parkinson's disease, but it does not slow down the disease or stop it from getting worse. It is taken by mouth. It can take two to three weeks of treatment before benefits are seen. Each dose then begins working in about ten minutes to two hours with a duration of effect of about five hours.

Dopaminergic means "related to dopamine" (literally, "working on dopamine"), dopamine being a common neurotransmitter. Dopaminergic substances or actions increase dopamine-related activity in the brain. Dopaminergic brain pathways facilitate dopamine-related activity. For example, certain proteins such as the dopamine transporter (DAT), vesicular monoamine transporter 2 (VMAT2), and dopamine receptors can be classified as dopaminergic, and neurons that synthesize or contain dopamine and synapses with dopamine receptors in them may also be labeled as dopaminergic. Enzymes that regulate the biosynthesis or metabolism of dopamine such as aromatic L-amino acid decarboxylase or DOPA decarboxylase, monoamine oxidase (MAO), and catechol O-methyl transferase (COMT) may be referred to as dopaminergic as well. Also, any endogenous or exogenous chemical substance that acts to affect dopamine receptors or dopamine release through indirect actions (for example, on neurons that synapse onto neurons that release dopamine or express dopamine receptors) can also be said to have dopaminergic effects, two prominent examples being opioids, which enhance dopamine release indirectly in the reward pathways, and some substituted amphetamines, which enhance dopamine release directly by binding to and inhibiting VMAT2.
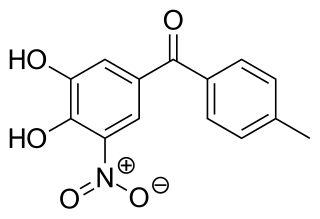
Tolcapone, sold under the brand name Tasmar, is a medication used to treat Parkinson's disease (PD). It is a selective, potent and reversible nitrocatechol-type inhibitor of the enzyme catechol-O-methyltransferase (COMT). It has demonstrated significant liver toxicity, which has led to suspension of marketing authorisations in a number of countries.

A dopamine agonist(DA) is a compound that activates dopamine receptors. There are two families of dopamine receptors, D1-like and D2-like. They are all G protein-coupled receptors. D1- and D5-receptors belong to the D1-like family and the D2-like family includes D2, D3 and D4 receptors. Dopamine agonists are primarily used in the treatment of the motor symptoms of Parkinson's disease, and to a lesser extent, in hyperprolactinemia and restless legs syndrome. They are also used off-label in the treatment of clinical depression. Impulse control disorders are associated with the use of dopamine agonists for whatever condition.
In the management of Parkinson's disease, due to the chronic nature of Parkinson's disease (PD), a broad-based program is needed that includes patient and family education, support-group services, general wellness maintenance, exercise, and nutrition. At present, no cure for the disease is known, but medications or surgery can provide relief from the symptoms.

A catechol-O-methyltransferase(COMT) inhibitor is a drug that inhibits the enzyme catechol-O-methyltransferase. This enzyme methylates catecholamines such as dopamine, norepinephrine and epinephrine. It also methylates levodopa. COMT inhibitors are indicated for the treatment of Parkinson's disease in combination with levodopa and an aromatic L-amino acid decarboxylase inhibitor. The therapeutic benefit of using a COMT inhibitor is based on its ability to prevent the methylation of levodopa to 3-O-methyldopa, thus increasing the bioavailability of levodopa. COMT inhibitors significantly decrease off time in people with Parkinson's disease also taking carbidopa/levodopa.

Droxidopa is a synthetic amino acid precursor which acts as a prodrug to the neurotransmitter norepinephrine (noradrenaline). Unlike norepinephrine, droxidopa is capable of crossing the protective blood–brain barrier (BBB).
Carbidopa/levodopa/entacapone, sold under the brand name Stalevo among others, is a dopaminergic fixed-dose combination medication that contains carbidopa, levodopa, and entacapone for the treatment of Parkinson's disease.

An aromatic L-amino acid decarboxylase inhibitor is a medication of type enzyme inhibitor which inhibits the synthesis of dopamine by the enzyme aromatic L-amino acid decarboxylase. It is used to inhibit the decarboxylation of L-DOPA to dopamine outside the brain, i.e. in the blood. This is primarily co-administered with L-DOPA to combat Parkinson's disease. Administration can prevent common side-effects, such as nausea and vomiting, as a result of interaction with D2 receptors in the vomiting center located outside the blood–brain barrier.

Safinamide is a drug used as an add-on treatment for Parkinson's disease with "off" episodes; it has multiple modes of action, including the inhibition of monoamine oxidase B.

Parkinson's disease (PD), or simply Parkinson's, is a long-term neurodegenerative disease of mainly the central nervous system that affects both the motor and non-motor systems of the body. The symptoms usually emerge slowly, and as the disease progresses, non-motor symptoms become more common. Usual symptoms include tremors, slowness of movement, rigidity, and difficulty with balance, collectively known as parkinsonism. Parkinson's disease dementia, falls and neuropsychiatric problems such as sleep abnormalities, psychosis, mood swings, or behavioral changes may arise in advanced stages as well.
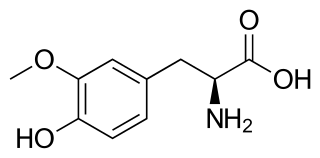
3-O-Methyldopa (3-OMD) is one of the most important metabolites of L-DOPA, a drug used in the treatment of the Parkinson's disease.
Peripherally selective drugs have their primary mechanism of action outside of the central nervous system (CNS), usually because they are excluded from the CNS by the blood–brain barrier. By being excluded from the CNS, drugs may act on the rest of the body without producing side-effects related to their effects on the brain or spinal cord. For example, most opioids cause sedation when given at a sufficiently high dose, but peripherally selective opioids can act on the rest of the body without entering the brain and are less likely to cause sedation. These peripherally selective opioids can be used as antidiarrheals, for instance loperamide (Imodium).
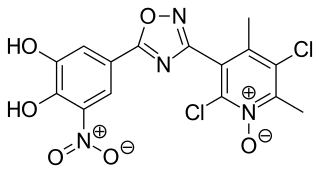
Opicapone, sold under the brand name Ongentys, is a medication which is administered together with levodopa in people with Parkinson's disease. Opicapone is a catechol-O-methyltransferase (COMT) inhibitor.