
A safety data sheet (SDS), material safety data sheet (MSDS), or product safety data sheet (PSDS) is a document that lists information relating to occupational safety and health for the use of various substances and products. SDSs are a widely used system for cataloging information on chemicals, chemical compounds, and chemical mixtures. SDS information may include instructions for the safe use and potential hazards associated with a particular material or product, along with spill-handling procedures. SDS formats can vary from source to source within a country depending on national requirements.
Cheminformatics is the use of computer and informational techniques applied to a range of problems in the field of chemistry. These in silico techniques are used, for example, in pharmaceutical companies and academic settings in the process of drug discovery. These methods can also be used in chemical and allied industries in various other forms.
Registry of Toxic Effects of Chemical Substances (RTECS) is a database of toxicity information compiled from the open scientific literature without reference to the validity or usefulness of the studies reported. Until 2001 it was maintained by US National Institute for Occupational Safety and Health (NIOSH) as a freely available publication. It is now maintained by the private company BIOVIA or from several value-added resellers and is available only for a fee or by subscription.

KEGG is a collection of databases dealing with genomes, biological pathways, diseases, drugs, and chemical substances. KEGG is utilized for bioinformatics research and education, including data analysis in genomics, metagenomics, metabolomics and other omics studies, modeling and simulation in systems biology, and translational research in drug development.
Chemical Entities of Biological Interest, also known as ChEBI, is a database and ontology of molecular entities focused on 'small' chemical compounds, that is part of the Open Biomedical Ontologies effort. The term "molecular entity" refers to any "constitutionally or isotopically distinct atom, molecule, ion, ion pair, radical, radical ion, complex, conformer, etc., identifiable as a separately distinguishable entity". The molecular entities in question are either products of nature or synthetic products which have potential bioactivity. Molecules directly encoded by the genome, such as nucleic acids, proteins and peptides derived from proteins by proteolytic cleavage, are not as a rule included in ChEBI.

ChemSpider is a database of chemicals. ChemSpider is owned by the Royal Society of Chemistry.

Canrenoic acid is a synthetic steroidal antimineralocorticoid which was never marketed.
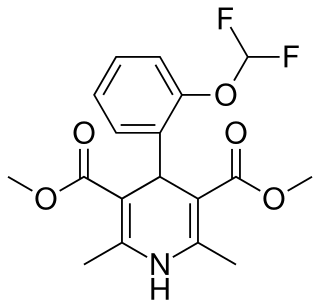
Riodipine (INN) is a calcium channel blocker.
Molecular communications systems use the presence or absence of a selected type of molecule to digitally encode messages. The molecules are delivered into communications media such as air and water for transmission. The technique also is not subject to the requirement of using antennas that are sized to a specific ratio of the wavelength of the signal. Molecular communication signals can be made biocompatible and require very little energy

Gestaclone, also known as 6-chloro-1α,2α:16α,17-bismethylene-4,6-pregnadiene-3,20-dione, is a steroidal progestin of unique chemical structure derived from progesterone that was first described in 1967 and was never marketed.

Leptacline (INN) is a drug described as a respiratory stimulant that was never marketed. It has a similar chemical structure to various piperidine and piperazine psychostimulants.
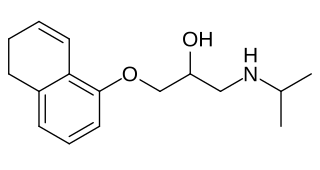
Idropranolol is a beta blocker that was never marketed.

Cloxestradiol acetate, also known as 17-(2,2,2-trichloroethoxy)estradiol O,O-diacetate, is a synthetic, steroidal estrogen derived from estradiol. It is the O,O-diacetate ester of cloxestradiol, which, in contrast to cloxestradiol acetate, was never marketed.

Doisynoestrol, also known as fenocycline, as well as cis-bisdehydrodoisynolic acid 7-methyl ether, is a synthetic nonsteroidal estrogen of the doisynolic acid group that is no longer marketed. It is a methyl ether of bisdehydrodoisynolic acid. Doisynoestrol was described in the literature in 1945.

Pentomone is a nonsteroidal antiandrogen (NSAA) described as a "prostate growth inhibitor" which was never marketed. It was synthesized and assayed in 1978.

Furostilbestrol (INN), also known as diethylstilbestrol di(2-furoate) or simply as diesthylstilbestrol difuroate, is a synthetic, nonsteroidal estrogen of the stilbestrol group related to diethylstilbestrol that was never marketed. It is an ester of diethylstilbestrol and was described in the literature in 1952.
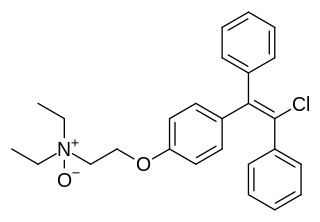
Clomifenoxide (INN), also known as clomifene N-oxide, is a nonsteroidal selective estrogen receptor modulator (SERM) of the triphenylethylene group that is described as an antiestrogen and "gonad stimulant" and was never marketed. It is an active metabolite of clomifene.

Nitromifene is a nonsteroidal selective estrogen receptor modulator (SERM) related to triphenylethylenes like tamoxifen that was never marketed. It is a mixture of (E)- and (Z)-isomers that possess similar antiestrogenic activity. The drug was described in 1966. Along with tamoxifen, nafoxidine, and clomifene, it was one of the earliest SERMs.

Timobesone is a synthetic glucocorticoid corticosteroid which was never marketed.

















