 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /ˌfludrəˈkɔːrtəzoʊn/ floo-drə-KOR-tih-zone |
| Trade names | Florinef, Astonin, others |
| Other names | StC-1400; 9α-Fluorohydrocortisone; 9α-Fluorocortisol; 9α-Fluoro-17α-hydroxycorticosterone; 9α-Fluoro-11β,17α,21-trihydroxypregn-4-ene-3,20-dione |
| AHFS/Drugs.com | Monograph |
| Routes of administration | By mouth |
| Drug class | Corticosteroid; glucocorticoid; mineralocorticoid |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Protein binding | High |
| Metabolism | Liver |
| Elimination half-life | 3.5 hours |
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| PubChemSID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| PDB ligand | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.004.395 |
| Chemical and physical data | |
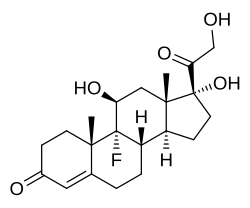
| Formula | C21H29FO5 |
| Molar mass | 380.456 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
 | |
| Clinical data | |
|---|---|
| Trade names | Cortineff, Florinef, Florinefe, Fludrocortison, others |
| Other names | Fluorohydrocortisone acetate; 9α-Fluorohydrocortisone 21-acetate; 9α-Fluoro-17α-hydroxycorticosterone 21-acetate; 9α-Fluoro-11β,17α,21-trihydroxypregn-4-ene-3,20-dione 21-acetate |
| Routes of administration | By mouth |
| Drug class | Corticosteroid; glucocorticoid; mineralocorticoid |
| Pharmacokinetic data | |
| Metabolism | Liver |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.004.395 |
| Chemical and physical data | |
| Formula | C23H31FO6 |
| Molar mass | 422.493 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 260 to 262 °C (500 to 504 °F) (dec.) |
| |
| |
Fludrocortisone, sold under the brand name Florinef among others, is a corticosteroid used to treat congenital adrenal hyperplasia, postural hypotension, and adrenal insufficiency. [3] [4] [5] In adrenal insufficiency, it is generally taken together with hydrocortisone. [5] Fludrocortisone is taken by mouth [5] and is most commonly used in its acetate form. [6]
Contents
- Medical uses
- Side effects
- Pharmacology
- Chemistry
- History
- Society and culture
- Generic name
- Brand names
- Availability
- References
Common side effects of fludrocortisone include high blood pressure, swelling, heart failure, and low blood potassium. [5] Other serious side effects can include low immune-system function, cataracts, muscle weakness, and mood changes. [5] Whether use of fludrocortisone during pregnancy is safe for the fetus is unknown. [7] Fludrocortisone is mostly a mineralocorticoid, but it also has glucocorticoid effects. [5]
Fludrocortisone was patented in 1953. [8] It is on the World Health Organization's List of Essential Medicines. [9]