Themes in bioinorganic chemistry
Several distinct systems are of identifiable in bioinorganic chemistry. Major areas include:
Metal ion transport and storage
A diverse collection of transporters (e.g. the ion pump NaKATPase), vacuoles, storage proteins (e.g. ferritin), and small molecules (e.g. siderophores) are employed to control metal ions concentration and bio-availability in living organisms. Crucially, many essential metals are not readily accessible to downstream proteins owing to low solubility in aqueous solutions or scarcity in the cellular environment. Organisms have developed a number of strategies for collecting and transporting such elements while limiting their cytotoxicity.
Enzymology
Many reactions in life sciences involve water and metal ions are often at the catalytic centers (active sites) for these enzymes, i.e. these are metalloproteins. Often the reacting water is a ligand (see metal aquo complex). Examples of hydrolase enzymes are carbonic anhydrase, metallophosphatases, and metalloproteinases. Bioinorganic chemists seek to understand and replicate the function of these metalloproteins.
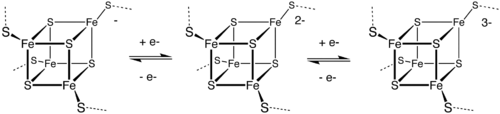
Metal-containing electron transfer proteins are also common. They can be organized into three major classes: iron–sulfur proteins (such as rubredoxins, ferredoxins, and Rieske proteins), blue copper proteins, and cytochromes. These electron transport proteins are complementary to the non-metal electron transporters nicotinamide adenine dinucleotide (NAD) and flavin adenine dinucleotide (FAD). The nitrogen cycle make extensive use of metals for the redox interconversions.
Toxicity
Several metal ions are toxic to humans and other animals. The bioinorganic chemistry of lead in the context of its toxicity has been reviewed. [4]
Oxygen transport and activation proteins
Aerobic life make extensive use of metals such as iron, copper, and manganese. Heme is utilized by red blood cells in the form of hemoglobin for oxygen transport and is perhaps the most recognized metal system in biology. Other oxygen transport systems include myoglobin, hemocyanin, and hemerythrin. Oxidases and oxygenases are metal systems found throughout nature that take advantage of oxygen to carry out important reactions such as energy generation in cytochrome c oxidase or small molecule oxidation in cytochrome P450 oxidases or methane monooxygenase. Some metalloproteins are designed to protect a biological system from the potentially harmful effects of oxygen and other reactive oxygen-containing molecules such as hydrogen peroxide. These systems include peroxidases, catalases, and superoxide dismutases. A complementary metalloprotein to those that react with oxygen is the oxygen evolving complex present in plants. This system is part of the complex protein machinery that produces oxygen as plants perform photosynthesis.
Bioorganometallic chemistry
Bioorganometallic systems feature metal-carbon bonds as structural elements or as intermediates. Bioorganometallic enzymes and proteins include the hydrogenases, FeMoco in nitrogenase, and methylcobalamin. These naturally occurring organometallic compounds. This area is more focused on the utilization of metals by unicellular organisms. Bioorganometallic compounds are significant in environmental chemistry. [5]

Metals in medicine
A number of drugs contain metals. This theme relies on the study of the design and mechanism of action of metal-containing pharmaceuticals, and compounds that interact with endogenous metal ions in enzyme active sites. The most widely used anti-cancer drug is cisplatin. MRI contrast agent commonly contain gadolinium. Lithium carbonate has been used to treat the manic phase of bipolar disorder. Gold antiarthritic drugs, e.g. auranofin have been commercialized. Carbon monoxide-releasing molecules are metal complexes have been developed to suppress inflammation by releasing small amounts of carbon monoxide. The cardiovascular and neuronal importance of nitric oxide has been examined, including the enzyme nitric oxide synthase. (See also: nitrogen assimilation.) Besides, metallic transition complexes based on triazolopyrimidines have been tested against several parasite strains. [6]
Environmental chemistry
Environmental chemistry traditionally emphasizes the interaction of heavy metals with organisms. Methylmercury has caused major disaster called Minamata disease. Arsenic poisoning is a widespread problem owing largely to arsenic contamination of groundwater, which affects many millions of people in developing countries. The metabolism of mercury- and arsenic-containing compounds involves cobalamin-based enzymes.
Biomineralization
Biomineralization is the process by which living organisms produce minerals, often to harden or stiffen existing tissues. Such tissues are called mineralized tissues. [7] [8] [9] Examples include silicates in algae and diatoms, carbonates in invertebrates, and calcium phosphates and carbonates in vertebrates. Other examples include copper, iron and gold deposits involving bacteria. Biologically-formed minerals often have special uses such as magnetic sensors in magnetotactic bacteria (Fe3O4), gravity sensing devices (CaCO3, CaSO4, BaSO4) and iron storage and mobilization (Fe2O3•H2O in the protein ferritin). Because extracellular [10] iron is strongly involved in inducing calcification, [11] [12] its control is essential in developing shells; the protein ferritin plays an important role in controlling the distribution of iron. [13]
